Gallopamil
Appearance
(Redirected from C28H40N2O5)
 | |
| Clinical data | |
|---|---|
| Other names | Methoxyverapamil |
| AHFS/Drugs.com | International Drug Names |
| ATC code | |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| UNII |
|
| KEGG | |
| ChEMBL | |
| CompTox Dashboard (EPA) | |
| Chemical and physical data | |
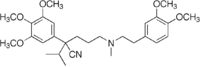
| Formula | C28H40N2O5 |
| Molar mass | 484.637 g·mol−1 |
| 3D model (JSmol) | |
| Chirality | Racemic mixture |
| |
| |
| | |
Gallopamil (INN) is an L-type calcium channel blocker that is an analog of verapamil. It is used in the treatment of abnormal heart rhythms.[1]
Synthesis[edit]

The alkylation between 3,4,5-Trimethoxyphenylacetonitrile [13338-63-1] (1)and 2-chloropropane [75-29-6] (2) gives 3-Methyl-2-(3,4,5-trimethoxyphenyl)butanenitrile [36622-33-0] (3). Treatment with N-Methyl-N-(3-Chloropropyl)Homoveratrylamine [36770-74-8] (4) completed the synthesis of Gallopamil (5).
References[edit]
- ^ Sewing KF, Hannemann H (1983). "Calcium channel antagonists verapamil and gallopamil are powerful inhibitors of acid secretion in isolated and enriched guinea pig parietal cells". Pharmacology. 27 (1): 9–14. doi:10.1159/000137824. PMID 6310646.
- ^ Mannhold, R.; Gallopamil Hydrochloride. Drugs Fut 1984, 9, 2, 108.
- ^ Theodore, Louis J.; Nelson, Wendel L. (1987). "Stereospecific synthesis of the enantiomers of verapamil and gallopamil". The Journal of Organic Chemistry. 52 (7): 1309–1315. doi:10.1021/jo00383a026.
- ^ https://caod.oriprobe.com/articles/28518495/Synthesis_of_Gallopamil_Hydrochloride.htm
- ^ Ferdinand Dengel, US4115432 (1978 to Knoll GmbH).
- ^ Werner Seitz, Klaus Scheib, & Alfred Michel, US4418017 (1983 to BASF SE).
- ^ Siegfried Herrling, US4305887 (1981 to BASF SE).
