Azelaic acid
| |

| |
| Names | |
|---|---|
| Preferred IUPAC name
Nonanedioic acid | |
| Identifiers | |
3D model (JSmol)
|
|
| 1101094 | |
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
| DrugBank | |
| ECHA InfoCard | 100.004.246 |
| EC Number |
|
| 261342 | |
| KEGG | |
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C9H16O4 | |
| Molar mass | 188.22 g/mol |
| Appearance | white solid |
| Density | 1.443 g/mL |
| Melting point | 109 to 111 °C (228 to 232 °F; 382 to 384 K)[1] |
| Boiling point | 286 °C (547 °F; 559 K) at 100 mmHg[1] |
| 2.14 g/L[2] | |
| Acidity (pKa) | 4.550, 5.498[2] |
| Pharmacology | |
| D10AX03 (WHO) | |
| Topical | |
| Pharmacokinetics: | |
| Very low | |
| 12 h | |
| Legal status | |
| Hazards | |
| GHS labelling: | |

| |
| Warning | |
| H315, H319 | |
| P264, P280, P302+P352, P305+P351+P338, P321, P332+P313, P337+P313, P362 | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
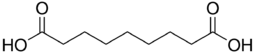
Azelaic acid (AzA) is an organic compound with the formula HOOC(CH2)7COOH.[3] This saturated dicarboxylic acid exists as a white powder. It is found in wheat, rye, and barley. It is a precursor to diverse industrial products including polymers and plasticizers, as well as being a component of a number of hair and skin conditioners.[4] AzA inhibits tyrosinase.[5]
Production
[edit]Azelaic acid is industrially produced by the ozonolysis of oleic acid. The side product is nonanoic acid. It is produced naturally by Malassezia furfur (also known as Pityrosporum ovale), a yeast that lives on normal skin. The bacterial degradation of nonanoic acid gives azelaic acid.[6]
Biological function
[edit]Plants biology
[edit]In plants, azelaic acid serves as a "distress flare" involved in defense responses after infection.[7] It serves as a signal that induces the accumulation of salicylic acid, an important component of a plant's defensive response.[8]
Human biology
[edit]The mechanism of action in humans is thought to be through the inhibition of hyperactive protease activity that converts cathelicidin into the antimicrobial skin peptide LL-37.[9]
Applications
[edit]
Polymers and related materials
[edit]Esters of this dicarboxylic acid find applications in lubrication and plasticizers. In lubricant industries it is used as a thickening agent in lithium complex grease. With hexamethylenediamine, azelaic acid forms Nylon-6,9, which finds specialized uses as a plastic.[4]
Medical
[edit]Azelaic acid is used to treat mild to moderate acne, both comedonal acne and inflammatory acne.[10][11] It belongs to a class of medication called dicarboxylic acids. It works by killing acne bacteria that infect skin pores. It also decreases the production of keratin, which is a natural substance that promotes the growth[clarification needed] of acne bacteria.[12] Azelaic acid is also used as a topical gel treatment for rosacea, due to its ability to reduce inflammation.[11] It clears the bumps and swelling caused by rosacea.
In topical pharmaceutical preparations and scientific research AzA is typically used in concentrations between 15% and 20% but some research demonstrates that in certain vehicle formulations the pharmaceutical effects of 10% Azelaic acid has the potential to be fully comparable to that of some 20% creams.[13]
Acne treatment
[edit]Azelaic acid is effective for mild to moderate acne when applied topically at a 15%-20% concentration.[14][15][16][17] In patients with moderate acne, twice daily application over 3 months of 20% AzA significantly reduced the number of comedones, papules, and pustules;[18][19] at this strength, it’s considered to be as effective as benzoyl peroxide 5%, tretinoin 0.05%, erythromycin 2%, and oral tetracycline at 500 mg-1000 mg.[20][21] In a comparative review of effects of topical AzA, Salicylic acid, Nicotinamide, Sulfur, Zinc, and alpha-hydroxy acid, AzA had more high-quality evidence of effectiveness than the rest.[22] Results can be expected after 4 weeks of twice-daily treatment. The effectiveness of long term use is unclear, but it’s been recommended that AzA be used for at least 6 months continuously for maintenance.[20]
Whitening agent
[edit]Azelaic acid is used for treatment of skin pigmentation, including melasma and postinflammatory hyperpigmentation, particularly in those with darker skin types. It has been recommended as an alternative to hydroquinone.[23] As a tyrosinase inhibitor,[5] azelaic acid reduces synthesis of melanin.[24] According to one report in 1988, azelaic acid in combination with zinc sulfate in vitro was found to be a potent (90% inhibition) 5α-reductase inhibitor, similar to the hair loss drugs finasteride and dutasteride.[25] In vitro research during mid-1980s evaluating azelaic acid's depigmenting (whitening) capability concluded it is effective (cytotoxic to melanocytes) at only high concentrations.[26]
A 1996 review claimed 20% AzA is as potent as 4% hydroquinone after a period of application of three months without the latter's adverse effects and even more effective if applied along with tretinoin for the same period of time.[27][19]
Brand names
[edit]Brand names for azelaic acid include Dermaz 99,[28] Crema Pella Perfetta (micronized azelaic acid, kojic dipalmitate, and liquorice extract), Azepur99, Azetec99, Azaclear (azelaic acid and niacinamide), AzClear Action, Azelex, White Action cream, Finacea, Finevin, Melazepam, Skinoren, Ezanic, Azelac, Azaderm, (Acnegen, Eziderm, Acnicam, Azelexin in Pakistan)[29]
References
[edit]- ^ a b Sigma-Aldrich catalog Archived April 9, 2008, at the Wayback Machine
- ^ a b Bretti, C.; Crea, F.; Foti, C.; Sammartano, S. (2006). "Solubility and Activity Coefficients of Acidic and Basic Nonelectrolytes in Aqueous Salt Solutions. 2. Solubility and Activity Coefficients of Suberic, Azelaic, and Sebacic Acids in NaCl(aq), (CH3)4NCl(aq), and (C2H5)4NI(aq) at Different Ionic Strengths and at t = 25 °C". J. Chem. Eng. Data. 51 (5): 1660–1667. doi:10.1021/je060132t.
- ^ Del Rosso, James Q. (2006-02-01). "The use of topical azelaic acid for common skin disorders other than inflammatory rosacea". Cutis. 77 (2 Suppl): 22–24. ISSN 0011-4162. PMID 16566285.
- ^ a b Cornils, Boy; Lappe, Peter (2006). "Dicarboxylic Acids, Aliphatic". Ullmann's Encyclopedia of Industrial Chemistry. Weinheim: Wiley-VCH. doi:10.1002/14356007.a08_523. ISBN 3527306730.
- ^ a b Pillaiyar, Thanigaimalai; Manickam, Manoj; Namasivayam, Vigneshwaran (2017-01-18). "Skin whitening agents: medicinal chemistry perspective of tyrosinase inhibitors". Journal of Enzyme Inhibition and Medicinal Chemistry. 32 (1): 403–425. doi:10.1080/14756366.2016.1256882. ISSN 1475-6366. PMC 6010116. PMID 28097901.
- ^ Integrative medicine. David Rakel (Fourth ed.). Philadelphia, PA. 2018. ISBN 978-0-323-35868-2. OCLC 982188831.
{{cite book}}: CS1 maint: location missing publisher (link) CS1 maint: others (link) - ^ Sarah Everts (January 31, 2011). "Vegetative Warfare". Chemical & Engineering News. 89 (5): 53–55. doi:10.1021/cen-v089n005.p053.
- ^ Jung, H. W.; Tschaplinski, T. J.; Wang, L.; Glazebrook, J.; Greenberg, J. T. (2009). "Priming in Systemic Plant Immunity". Science. 324 (5923): 89–91. Bibcode:2009Sci...324...89W. doi:10.1126/science.1170025. PMID 19342588. S2CID 206518245.
- ^ Reinholz, M.; Ruzicka, T.; Schauber, J. (2012). "Cathelicidin LL-37: An Antimicrobial Peptide with a Role in Inflammatory Skin Disease". Annals of Dermatology. 24 (2): 126–135. doi:10.5021/ad.2012.24.2.126. PMC 3346901. PMID 22577261.
- ^ "Azelaic Acid for Acne". WebMD.
- ^ a b "Azelaic acid topical". Drugs.com.
- ^ Liu, R. H.; Smith, M. K.; Basta, S. A.; Farmer, E. R. (2006). "Azelaic acid in the treatment of papulopustular rosacea – A systematic review of randomized controlled trials". Archives of Dermatology. 142 (8): 1047–1052. doi:10.1001/archderm.142.8.1047. PMID 16924055.
- ^ Burchacka, E.; Potaczek, P.; Paduszyński, P.; Karłowicz-Bodalska, K.; Han, T.; Han, S. (2016-10-01). "New effective azelaic acid liposomal gel formulation of enhanced pharmaceutical bioavailability". Biomedicine & Pharmacotherapy. 83: 771–775. doi:10.1016/j.biopha.2016.07.014. ISSN 0753-3322. PMID 27484346.
- ^ Sieber MA, Hegel JK (November 2013). "Azelaic acid: Properties and mode of action". Skin Pharmacology and Physiology (Review). 27 Suppl 1 (Supplement 1): 9–17. doi:10.1159/000354888. PMID 24280644. S2CID 8848543.
- ^ Pugashetti R, Shinkai K (July 2013). "Treatment of acne vulgaris in pregnant patients". Dermatologic Therapy (Review). 26 (4): 302–11. doi:10.1111/dth.12077. PMID 23914887. S2CID 5750718.
- ^ Gollnick, H. P.; Graupe, K.; Zaumseil, R. P. (2004). "15% Azelainsauregel in der Behandlung der Akne. Zwei doppelblinde klinische Vergleichsstudien" [Azelaic acid 15% gel in the treatment of acne vulgaris. Combined results of two double-blind clinical comparative studies]. Journal der Deutschen Dermatologischen Gesellschaft. 2 (10): 841–847. doi:10.1046/j.1439-0353.2004.04731.x. PMID 16281587. S2CID 58809558. Retrieved 2021-05-14.
- ^ Thiboutot, Diane (January 2008). "Versatility of azelaic acid 15% gel in treatment of inflammatory acne vulgaris". Journal of Drugs in Dermatology. 7 (1): 13–16. ISSN 1545-9616. PMID 18246693.
- ^ "Azelaic Acid And Its Benefits". Chemist at Play. Retrieved 2022-03-10.
- ^ a b Fitton, A.; Goa, K. L. (May 1991). "Azelaic acid. A review of its pharmacological properties and therapeutic efficacy in acne and hyperpigmentary skin disorders". Drugs. 41 (5): 780–798. doi:10.2165/00003495-199141050-00007. ISSN 0012-6667. PMID 1712709. S2CID 46960738.
- ^ a b Morelli V, Calmet E, Jhingade V (June 2010). "Alternative therapies for common dermatologic disorders, part 2". Primary Care (Review). 37 (2): 285–96. doi:10.1016/j.pop.2010.02.005. PMID 20493337.
- ^ Woolery-Lloyd, Heather C.; Keri, Jonette; Doig, Stefan (2013-04-01). "Retinoids and azelaic acid to treat acne and hyperpigmentation in skin of color". Journal of Drugs in Dermatology. 12 (4): 434–437. ISSN 1545-9616. PMID 23652891.
- ^ Liu, Haibo; Yu, Haiyan; Xia, Jun; Liu, Ling; Liu, Guanjian; Sang, Hong; Peinemann, Frank (2020-11-01). "Evidence-based topical treatments (azelaic acid, salicylic acid, nicotinamide, sulfur, zinc, and fruit acid) for acne: an abridged version of a Cochrane systematic review". Journal of Evidence-Based Medicine. 13 (4): 275–283. doi:10.1111/jebm.12411. ISSN 1756-5391. PMID 33034949. S2CID 222236883.
- ^ Draelos, Z. (Sep–Oct 2007). "Skin lightening preparations and the hydroquinone controversy". Dermatologic Therapy. 20 (5): 308–313. doi:10.1111/j.1529-8019.2007.00144.x. PMID 18045355. S2CID 24913995.
- ^ Grimes, Pearl E. (2007-07-01). Aesthetics and Cosmetic Surgery for Darker Skin Types. Lippincott Williams & Wilkins. pp. 74 ff. ISBN 978-0-7817-8403-0. Retrieved 9 August 2011.
- ^ Stamatiadis, D; Bulteau-Portois, Marie-Claire; Mowszowicz, Irene (1988). "Inhibition of 5α-reductase activity in human skin by zinc and azelaic acid". British Journal of Dermatology. 119 (5): 627–32. doi:10.1111/j.1365-2133.1988.tb03474.x. PMID 3207614. S2CID 28506969.
- ^ Pathak, M. A.; Ciganek, E. R.; Wick, M.; Sober, A. J.; Farinelli, W. A.; Fitzpatrick, T. B. (September 1985). "An evaluation of the effectiveness of azelaic acid as a depigmenting and chemotherapeutic agent". The Journal of Investigative Dermatology. 85 (3): 222–228. doi:10.1111/1523-1747.ep12276684. ISSN 0022-202X. PMID 4031538.
- ^ Breathnach, A. S. (1996-01-01). "Melanin hyperpigmentation of skin: melasma, topical treatment with azelaic acid, and other therapies". Cutis. 57 (1 Suppl): 36–45. ISSN 0011-4162. PMID 8654129.
- ^ https://pharmaceutical.basf.com/en/APIs-Raw-Materials/Dermaz.html[full citation needed][permanent dead link]
- ^ "Azelaic Acid brands in Pakistan". www.druginfosys.com. Retrieved 2021-04-17.
