Kupffer cell
| Kupffer cell | |
|---|---|
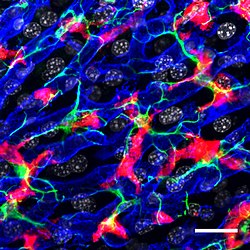 Confocal microscopy picture showing the steady-state location and interactions between Kupffer cells (Red), hepatic stellate cells (green) and liver sinusoidal endothelial cells (blue). Cell nuclei are in grey.[1] | |
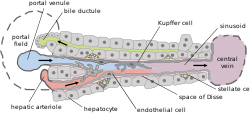 Basic liver structure | |
| Details | |
| Location | Liver |
| Function | Macrophage |
| Identifiers | |
| Latin | macrophagocytus stellatus |
| MeSH | D007728 |
| TH | H3.04.05.0.00016 |
| FMA | 14656 |
| Anatomical terms of microanatomy | |
Kupffer cells, also known as stellate macrophages and Kupffer–Browicz cells, are specialized cells localized in the liver within the lumen of the liver sinusoids and are adhesive to their endothelial cells which make up the blood vessel walls. Kupffer cells comprise the largest population of tissue-resident macrophages in the body. Gut bacteria, bacterial endotoxins, and microbial debris transported to the liver from the gastrointestinal tract via the portal vein will first come in contact with Kupffer cells, the first immune cells in the liver. It is because of this that any change to Kupffer cell functions can be connected to various liver diseases such as alcoholic liver disease, viral hepatitis, intrahepatic cholestasis, steatohepatitis, activation or rejection of the liver during liver transplantation and liver fibrosis.[2][3] They form part of the mononuclear phagocyte system.
Location and structure
[edit]Kupffer cells can be found attached to sinusoidal endothelial cells in both the centrilobular and periportal regions of the hepatic lobules. Kupffer cell function and structures are specialized depending on their location. Periportal Kupffer cells tend to be larger and have more lysosomal enzyme and phagocytic activity, whereas centrilobular Kupffer cells create more superoxide radical.
Kupffer cells are amoeboid in character, with surface features including microvilli, pseudopodia and lamellipodia, which project in every direction. The microvilli and pseudopodia play a role in the endocytosis of particles.
The nucleus is indented and ovoid, and can be lobulated.
Notable cytoplasmic elements include ribosomes, Golgi complexes, centrioles, microtubules and microfilaments. Kupffer cells also contain rough endoplasmic reticulum, a nuclear envelope, and annulate lamellae, all of which demonstrate peroxidase activity.
Importantly, Kupffer cells express the SR-AI/II scavenger receptor. This receptor is involved in recognising and binding the lipid A domain of lipopolysaccharide (LPS) and lipoteichoic acid. (Lipopolysaccharide (LPS) is a bacterial endotoxin which is found in the cell wall gram-negative bacteria, whereas lipoteichoic acid is present in gram-positive bacteria.) Because of this detection system, Kupffer cells play a critical role in initiating and mediating immune responses to bacterial infection of the liver.[4]
Development
[edit]Development of an initial population of Kupffer cells begins in the embryonic yolk sac where precursor cells differentiate into fetal macrophages. Once they enter the blood stream, they migrate to the fetal liver where they stay. There they complete their differentiation into Kupffer cells. Under normal conditions, these Kupffer cell populations are long-lived and self-renewing.[5]
However, if resident Kupffer cell populations are depleted, monocytes derived from hematopoietic stem cells in the bone marrow and transported through blood circulation to the liver can also fully differentiate into true Kupffer cells. Unlike other tissue macrophages, which must be continually renewed by circulating monocytes, these monocyte-derived Kupffer cells are capable of self-renewal once a population is established.[6]
Development of mature Kupffer cells is regulated by numerous growth factors, with macrophage colony-stimulating factor (CSF1) playing a key role. Cytokines involved in type 2 inflammation, such as IL-4, may also stimulate Kupffer cell proliferation. A time frame of 14 to 21 days for complete replenishment of Kupffer cell populations has been demonstrated in animal studies. Despite high monocyte influx and maturation rates, hepatic Kupffer cell populations are tightly maintained. Evidently, there is a high rate of turnover, with the average lifespan of a Kupffer cell estimated at 3.8 days. However, the ultimate fate of Kupffer cells in vivo is not yet fully understood.[4][7][3]
Function
[edit]The primary function of the Kupffer cell is to remove foreign debris and particles that have come from the hepatic portal system when passing through the liver. It is possible for the Kupffer cells to take in large particles by phagocytosis and smaller particles via pinocytosis.[4] Kupffer cells are integral in the innate responses of the immune system. They are important for host defense and play a role in the metabolism of many different compounds including, lipids, protein complexes and small particles. They are also useful in removing apoptotic cells from circulation.[2][3]
The amount of Kupffer cells in the liver is held constant. Kupffer cells have a proliferative capacity, allowing for cell populations to replenish themselves: this is in complete contrast to monocyte-derived macrophages that have no proliferative potential. Old or defective cells are removed through apoptosis, as well as through being phagocytized by neighbouring Kupffer cells.
Kupffer cells are heterogeneous in their function, dependent on their location in the liver lobules. Cells in the periportal zone are directly exposed to bloodflow, and express greater lysosomal activity to more efficiently process incoming foreign substances. In contrast, cells in the centrilobular zone experience less perfusion, and are equipped with greater stores of superoxide to combat deeply-penetrating injuries and infections.
In response to infection or irritation, Kupffer cells can produce inflammatory cytokines, TNF-alpha, oxygen radicals, and proteases. Excessive production of these mediators is linked to the development of liver injury.[4]
Apart from clearing bacteria, Kupffer cells are also responsible for recycling hemoglobin by destroying senescent red blood cells through phagocytic action. The globin chains are re-used, while the iron-containing portion, heme, is further broken down into iron, which is re-used, and bilirubin, which is conjugated to glucuronic acid within hepatocytes and secreted into the bile.
Helmy et al. identified a receptor present in Kupffer cells, the complement receptor of the immunoglobulin family (CRIg). Mice without CRIg could not clear complement system-coated pathogens. CRIg is conserved in mice and humans and is a critical component of the innate immune system.[8]
Clinical significance
[edit]Kupffer cells are incredibly plastic cells that have the capability to polarize specific activation states and can perform different functions in different microenvironments. M1 (classical activation) and M2 (alternative activation) designate the two extremes of macrophage polarization. M1-polarized Kupffer cells produce a large amount of pro-inflammatory cytokines like TNF-alpha. On the other hand, M2-polarized Kupffer cells produce a large quantity of anti-inflammatory mediators, for example, IL-10.[9][4]
Kupffer cells play a role in the pathogenesis of a damaged liver in response to sepsis. The macrophages in the liver activate and release both IL-1 and TNF-alpha. In turn, this activates leukocytes and sinusoidal endothelial cells to express ICAM-1. This results in tissue damage to the endothelium because of proteases, oxygen radicals, prostanoids and other substances from leukocytes.
Kupffer cell activation contributes to pathogenesis of both chronic and acute alcoholic liver disease in response to ethanol-induced liver injury, common in chronic alcoholics. Chronic alcoholism and liver injury deal with a two-hit system. While the first hit is direct, mediated by the direct toxicity of ethanol and its metabolic byproducts, the second hit is indirect, mediated by increased uptake of lipopolysaccharide (endotoxin) from the intestine.
Ethanol increases permeability of the intestinal epithelium, resulting in endotoxin produced by the intestinal flora leaking from the intestinal lumen into the liver via the portal vein. The presence of endotoxin induces a strong M1 polarization of Kupffer cells. A large amount of reactive oxygen species, pro-inflammatory cytokines and chemokines are produced by the activated Kupffer cells which lead to liver injury.
The cascade begins with endotoxin-mediated activation of the Toll-like receptor 4 (TLR4) and CD14, receptors on the Kupffer cell that internalize endotoxin. This in turn activates the transcription of pro-inflammatory cytokines and tumor necrosis factor-alpha (TNFα), with concurrent production of superoxides.
Cytokines and superoxides go on to cause inflammation and oxidizing damage respectively, while TNFα triggers the stellate cells in the liver to initiate collagen synthesis. These processes result in fibrosis, or scarring of the liver. Fibrosis will eventually cause cirrhosis, a loss of function of the liver due to extensive scarring.[10]
History
[edit]The cells were first observed by Karl Wilhelm von Kupffer in 1876.[11] The scientist called them "Sternzellen" (star cells or hepatic stellate cell) but thought, inaccurately, that they were an integral part of the endothelium of the liver blood vessels and that they originated from it. In 1898, after several years of research, Tadeusz Browicz identified them, correctly, as macrophages.[12][13][14][15]
References
[edit]- ^ Bonnardel J, T'Jonck W, Gaublomme D, Browaeys R, Scott CL, Martens L, et al. (October 2019). "Stellate Cells, Hepatocytes, and Endothelial Cells Imprint the Kupffer Cell Identity on Monocytes Colonizing the Liver Macrophage Niche". Immunity. 51 (4): 638–654.e9. doi:10.1016/j.immuni.2019.08.017. PMC 6876284. PMID 31561945.
- ^ a b Nguyen-Lefebvre, Anh Thu; Horuzsko, Anatolij (2015). "Kupffer Cell Metabolism and Function". Journal of Enzymology and Metabolism. 1 (1). PMC 4771376. PMID 26937490.
- ^ a b c Dixon, Laura J.; Barnes, Mark; Tang, Hui; Pritchard, Michele T.; Nagy, Laura E. (April 2013). "Kupffer Cells in the Liver". Comprehensive Physiology. 3 (2): 785–797. doi:10.1002/cphy.c120026. ISSN 2040-4603. PMC 4748178. PMID 23720329.
- ^ a b c d e Basit, Hajira; Tan, Michael L.; Webster, Daniel R. (2020), "Histology, Kupffer Cell", StatPearls, Treasure Island (FL): StatPearls Publishing, PMID 29630278, retrieved 2020-08-25
- ^ Naito M, Hasegawa G, Takahashi K (November 1997). "Development, differentiation, and maturation of Kupffer cells". Microscopy Research and Technique. 39 (4): 350–64. doi:10.1002/(SICI)1097-0029(19971115)39:4<350::AID-JEMT5>3.0.CO;2-L. PMID 9407545. S2CID 21193303.
- ^ Scott C, Zheng F, De Baetselier P, et al. (27 January 2016). "Bone marrow-derived monocytes give rise to self-renewing and fully differentiated Kupffer cells". Nature Communications. 7, 10321 (2016): 10321. Bibcode:2016NatCo...710321S. doi:10.1038/ncomms10321. PMC 4737801. PMID 26813785.
- ^ Chen, Jiajia; Deng, Xiaoyi; Liu, Yongjian; Tan, Qiuhua; Huang, Guidong; Che, Qishi; Guo, Jiao; Su, Zhengquan (2020). "Kupffer Cells in Non-alcoholic Fatty Liver Disease: Friend or Foe?". International Journal of Biological Sciences. 16 (13): 2367–2378. doi:10.7150/ijbs.47143. PMC 7378652. PMID 32760204. Retrieved 2020-08-31.
- ^ Helmy KY, Katschke KJ, Gorgani NN, Kljavin NM, Elliott JM, Diehl L, et al. (March 2006). "CRIg: a macrophage complement receptor required for phagocytosis of circulating pathogens". Cell. 124 (5): 915–27. doi:10.1016/j.cell.2005.12.039. PMID 16530040. S2CID 15525209.
- ^ Zeng, Tao; Zhang, Cui-Li; Xiao, Mo; Yang, Rui; Xie, Ke-Qin (2016). "Critical Roles of Kupffer Cells in the Pathogenesis of Alcoholic Liver Disease: From Basic Science to Clinical Trials". Frontiers in Immunology. 7: 538. doi:10.3389/fimmu.2016.00538. ISSN 1664-3224. PMC 5126119. PMID 27965666.
- ^ Wheeler MD (2003). "Endotoxin and Kupffer cell activation in alcoholic liver disease". Alcohol Research & Health. 27 (4): 300–6. PMC 6668869. PMID 15540801.
- ^ Haubrich WS (July 2004). "Kupffer of Kupffer cells". Gastroenterology. 127 (1): 16. doi:10.1053/j.gastro.2004.05.041. PMID 15236167.
- ^ Szymańska R, Schmidt-Pospuła M (1979). "[Studies of liver's reticuloendothelial cells by Tadeusz Browicz and Karl Kupffer. A historical outline]". Archiwum Historii Medycyny. 42 (3): 331–6. PMID 386989.
- ^ Stachura J, Gałązka K (December 2003). "History and current status of Polish gastroenterological pathology". Journal of Physiology and Pharmacology. 54 Suppl 3: 183–92. PMID 15075472.
- ^ Śródka A, Gryglewski RW, Szczepański W (2006). "Browicz or Kupffer cells?". Polish Journal of Pathology. 57 (4): 183–5. PMID 17285760.
- ^ Wake K (2009). "[Browicz's phagocytic cells and Kupffer's 'so-called stellate cells']". Kaibogaku Zasshi. 84 (1): 17–21. PMID 19413196.
External links
[edit]- Anatomy photo: digestive/mammal/liver5/liver4 - Comparative Organology at University of California, Davis - "Mammal, liver (EM, Low)"
- Histology image: 15508loa – Histology Learning System at Boston University
