Osmium(IV) chloride
| |
| Names | |
|---|---|
| IUPAC name
Osmium(IV) chloride
| |
| Other names
Osmium chloride, osmium tetrachloride
| |
| Identifiers | |
3D model (JSmol)
|
|
| ECHA InfoCard | 100.151.226 |
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| OsCl4 | |
| Molar mass | 332.041 g/mol |
| Appearance | red-black orthorhombic crystals |
| Density | 4.38 g/cm3 |
| Melting point | decomposes at 323°C |
| reacts with water | |
| Solubility | soluble in hydrochloric acid |
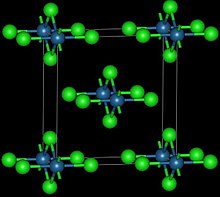
| Structure | |
| Orthorhombic, oS10 | |
| Cmmm, No. 65 | |
| Related compounds | |
Other anions
|
Osmium tetrabromide |
Other cations
|
Iron(III) chloride Ruthenium(III) chloride Osmium(III) chloride |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Osmium(IV) chloride or osmium tetrachloride is the inorganic compound composed of osmium and chlorine with the empirical formula OsCl4. It exists in two polymorphs (crystalline forms). The compound is used to prepare other osmium complexes.
Preparation, structure, reactions
[edit]It was first reported in 1909 as the product of chlorination of osmium metal.[1] This route affords the high temperature polymorph:[2]
- Os + 2 Cl2 → OsCl4
This reddish-black polymorph is orthorhombic and adopts a structure in which osmium centres are octahedrally coordinated, sharing opposite edges of the OsCl6 octahedra to form a chain.[3] A brown, apparently cubic polymorph forms upon reduction of osmium tetroxide with thionyl chloride:[4]
- OsO4 + 2 SOCl2 → OsCl4 + 2 Cl2 + 2 SO2
Osmium tetraoxide dissolves in hydrochloric acid to give the hexachloroosmate anion:
- OsO4 + 10 HCl → H2OsCl6 + 2 Cl2 + 4 H2O
References
[edit]- ^ Otto Ruff and Ferd. Bornemann (1910). "Über das Osmium, seine analytische Bestimmung, seine Oxyde und seine Chloride". Zeitschrift für anorganische Chemie. 65: 429–456. doi:10.1002/zaac.19100650126.
- ^ Cotton, S. A. (1997). Chemistry of Precious Metals. London: Chapman and Hall. ISBN 0-7514-0413-6.
- ^ Wells A.F. (1984). Structural Inorganic Chemistry (5th ed.). Oxford Science Publications. ISBN 0-19-855370-6.
- ^ Paul Machmer (1967). "On the polymorphism of osmium tetrachloride". Chem. Commun. (12): 610a. doi:10.1039/C1967000610A.
