Glutamate-sensitive fluorescent reporter
A genetically engineered fluorescent protein that changes its fluorescence when bound to the neurotransmitter glutamate.[1] Glutamate-sensitive fluorescent reporters (iGluSnFR, colloquially pronounced 'glue sniffer') are used to monitor the activity of presynaptic terminals by fluorescence microscopy. GluSnFRs are a class of optogenetic sensors used in neuroscience research.[2] In brain tissue, two-photon microscopy is typically used to monitor GluSnFR fluorescence.
Design[edit]
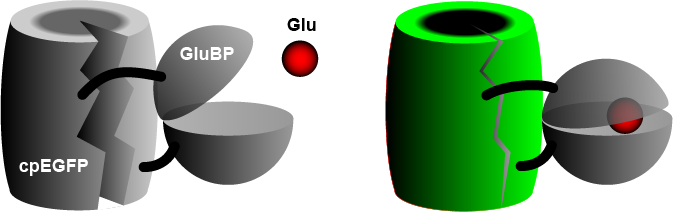
The widely used iGluSnFR consists of a circularly permuted enhanced green fluorescent protein (cpEGFP) fused to a glutamate binding protein (GluBP) from a bacterium.[3] When GluBP binds a glutamate molecule, it changes its shape, pulling the EGFP barrel together, increasing fluorescence. A specific peptide segment (PDGFR) is included to bring the sensor to the outside of the cell membrane.[4] In the more recent version by Aggarwal et al. (2022),[1] researchers introduced iGluSnFR to two additional anchoring domains, a glycosylphostidylinositol (GPI) anchor, and a modified form of the cytosolic -cterminal domain of Stargazin with a PDZ ligand.
History[edit]
The first genetically encoded fluorescent glutamate sensors (FLIPE, GluSnFR and SuperGluSnFR) were constructed by attaching cyan-fluorescent protein (CFP) and yellow-fluorescent protein (YFP) to a bacterial glutamate binding protein (GluBP).[5][6] Glutamate binding changed the distance between CFP and YFP, changing the efficiency of energy transfer (FRET) between the two fluorophores.[7][8] A breakthrough in visualizing glutamate release was achieved with iGluSnFR, a single-fluorophore glutamate sensor based on EGFP producing a ~5‑fold increase in fluorescence.[3] To measure synaptic transmission at high frequencies, novel iGluSnFR variants with accelerated kinetics have recently been developed.[9][10]
References[edit]
- ^ a b Aggarwal, Abhi; Liu, Rui; Chen, Yang; Ralowicz, Amelia J.; Bergerson, Samuel J.; Tomaska, Filip; Hanson, Timothy L.; Hasseman, Jeremy P.; Reep, Daniel; Tsegaye, Getahun; Yao, Pantong; Ji, Xiang; Kloos, Marinus; Walpita, Deepika; Patel, Ronak (2022-02-15). "Glutamate indicators with improved activation kinetics and localization for imaging synaptic transmission": 2022.02.13.480251. doi:10.1101/2022.02.13.480251v1.
{{cite journal}}: Cite journal requires|journal=(help) - ^ Hefendehl, J. K.; LeDue, J.; Ko, R. W. Y.; Mahler, J.; Murphy, T. H.; MacVicar, B. A. (2016-11-11). "Mapping synaptic glutamate transporter dysfunction in vivo to regions surrounding Aβ plaques by iGluSnFR two-photon imaging". Nature Communications. 7: 13441. Bibcode:2016NatCo...713441H. doi:10.1038/ncomms13441. PMC 5114608. PMID 27834383.
- ^ a b Marvin, Jonathan S; Borghuis, Bart G; Tian, Lin; Cichon, Joseph; Harnett, Mark T; Akerboom, Jasper; Gordus, Andrew; Renninger, Sabine L; Chen, Tsai-Wen (2013). "An optimized fluorescent probe for visualizing glutamate neurotransmission". Nature Methods. 10 (2): 162–170. doi:10.1038/nmeth.2333. ISSN 1548-7105. PMC 4469972. PMID 23314171.
- ^ Marvin, Jonathan S.; Schreiter, Eric R.; Echevarría, Ileabett M.; Looger, Loren L. (2011-11-01). "A genetically encoded, high-signal-to-noise maltose sensor". Proteins: Structure, Function, and Bioinformatics. 79 (11): 3025–3036. doi:10.1002/prot.23118. ISSN 1097-0134. PMC 3265398. PMID 21989929.
- ^ Hu, Yonglin; Fan, Cheng-Peng; Fu, Guangsen; Zhu, Deyu; Jin, Qi; Wang, Da-Cheng (2008). "Crystal Structure of a Glutamate/Aspartate Binding Protein Complexed with a Glutamate Molecule: Structural Basis of Ligand Specificity at Atomic Resolution". Journal of Molecular Biology. 382 (1): 99–111. doi:10.1016/j.jmb.2008.06.091. PMID 18640128.
- ^ De Lorimier, Robert M.; Smith, J. Jeff; Dwyer, Mary A.; Looger, Loren L.; Sali, Kevin M.; Paavola, Chad D.; Rizk, Shahir S.; Sadigov, Shamil; Conrad, David W. (2002-11-01). "Construction of a fluorescent biosensor family". Protein Science. 11 (11): 2655–2675. doi:10.1110/ps.021860. ISSN 1469-896X. PMC 2373719. PMID 12381848.
- ^ Okumoto, Sakiko; Looger, Loren L.; Micheva, Kristina D.; Reimer, Richard J.; Smith, Stephen J.; Frommer, Wolf B. (2005-06-14). "Detection of glutamate release from neurons by genetically encoded surface-displayed FRET nanosensors". Proceedings of the National Academy of Sciences of the United States of America. 102 (24): 8740–8745. Bibcode:2005PNAS..102.8740O. doi:10.1073/pnas.0503274102. ISSN 0027-8424. PMC 1143584. PMID 15939876.
- ^ Hires, Samuel Andrew; Zhu, Yongling; Tsien, Roger Y. (2008-03-18). "Optical measurement of synaptic glutamate spillover and reuptake by linker optimized glutamate-sensitive fluorescent reporters". Proceedings of the National Academy of Sciences. 105 (11): 4411–4416. Bibcode:2008PNAS..105.4411H. doi:10.1073/pnas.0712008105. ISSN 0027-8424. PMC 2393813. PMID 18332427.
- ^ Helassa, Nordine; Dürst, Céline D.; Coates, Catherine; Kerruth, Silke; Arif, Urwa; Schulze, Christian; Wiegert, J. Simon; Geeves, Michael; Oertner, Thomas G.; Török, Katalin (2018-05-22). "Ultrafast glutamate sensors resolve high-frequency release at Schaffer collateral synapses". Proceedings of the National Academy of Sciences. 115 (21): 5594–5599. doi:10.1073/pnas.1720648115. PMC 6003469. PMID 29735711.
- ^ Marvin, Jonathan S.; Scholl, Benjamin; Wilson, Daniel E.; Podgorski, Kaspar; Kazemipour, Abbas; Müller, Johannes Alexander; Schoch, Susanne; Quiroz, Francisco José Urra; Rebola, Nelson; Bao, Huan; Little, Justin P. (November 2018). "Stability, affinity, and chromatic variants of the glutamate sensor iGluSnFR". Nature Methods. 15 (11): 936–939. doi:10.1038/s41592-018-0171-3. ISSN 1548-7105. PMC 6394230. PMID 30377363.
