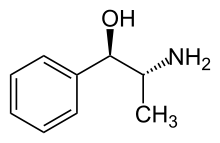
L-Norpseudoephedrine
 | |
| Clinical data | |
|---|---|
| ATC code |
|
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| UNII | |
| CompTox Dashboard (EPA) | |
| ECHA InfoCard | 100.164.234 |
| Chemical and physical data | |
| Formula | C9H13NO |
| Molar mass | 151.209 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
L-Norpseudoephedrine, or (−)-norpseudoephedrine, is a psychostimulant drug of the amphetamine family. It is one of the four optical isomers of phenylpropanolamine, the other three being cathine ((+)-norpseudoephedrine), (−)-norephedrine, and (+)-norephedrine; as well as one of the two enantiomers of norpseudoephedrine (the other being cathine).[1] Similarly to cathine, L-norpseudoephedrine acts as a releasing agent of norepinephrine (EC50 = 30 nM) and to a lesser extent of dopamine (EC50 = 294 nM).[2] Due to the 10-fold difference in its potency for inducing the release of the two neurotransmitters however, L-norpseudoephedrine could be called a modestly selective or preferential norepinephrine releasing agent, similarly to related compounds like ephedrine and pseudoephedrine.
See also
[edit]References
[edit]- ^ Macdonald F (1997). Dictionary of Pharmacological Agents. CRC Press. p. 121. ISBN 978-0-412-46630-4. Retrieved 18 May 2012.
- ^ Rothman RB, Vu N, Partilla JS, Roth BL, Hufeisen SJ, Compton-Toth BA, et al. (October 2003). "In vitro characterization of ephedrine-related stereoisomers at biogenic amine transporters and the receptorome reveals selective actions as norepinephrine transporter substrates". The Journal of Pharmacology and Experimental Therapeutics. 307 (1): 138–45. doi:10.1124/jpet.103.053975. PMID 12954796. S2CID 19015584.
| Phenethylamines |
|
|---|---|
| Amphetamines |
|
| Phentermines |
|
| Cathinones |
|
| Phenylisobutylamines | |
| Phenylalkylpyrrolidines | |
| Catecholamines (and close relatives) |
|
| Miscellaneous |
|
- Articles with short description
- Short description matches Wikidata
- ECHA InfoCard ID from Wikidata
- Infobox drug articles with non-default infobox title
- Articles without EBI source
- Chemical pages without DrugBank identifier
- Articles without KEGG source
- Drugs with no legal status
- Articles containing unverified chemical infoboxes
