Nucleocidin
| |
| Names | |
|---|---|
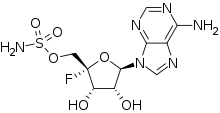
| IUPAC name
4′-Fluoroadenosine 5′-sulfamate
| |
| Systematic IUPAC name
[(2S,3S,4R,5R)-5-(6-Amino-9H-purin-9-yl)-2-fluoro-3,4-dihydroxyoxan-2-yl]methyl sulfamate | |
| Identifiers | |
3D model (JSmol)
|
|
| ChEBI | |
| ChemSpider | |
PubChem CID
|
|
| UNII | |
| |
| |
| Properties | |
| C10H13FN6O6S | |
| Molar mass | 364.31 g·mol−1 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Nucleocidin is a fluorine-containing nucleoside produced by Streptomyces calvus.[1]
Chemical structure
[edit]Nucleocidin stems from the ribonucleoside adenosine[2] - is unique because it possess two rare functional groups: a fluorine atom and a sulfamyl ester [3]
During 1968 the attempts to identify nucleocidin were made and at that time it was assigned to a structure of a 9-adenyl-4' -sulfamoyloxypentofuranoside, which was mainly based on experiments from NMR and mass spectrometry, as well as testing in chemical reactions. It was ultimately proven to have a structure of a 4' -fluoro-5' -O-sulphamoyladenosine.[4]
Microbiological origin
[edit]Nucleocidin is an antibiotic produced from Streptomyces calvus. Though toxic to mammals, it is able to function against bacteria both gram negative gram positive. It may be used against trypanosomes.[5]
Though commonly known to be produced by Streptomyces calvus, nucleocidin is produced in greater yield by Streptomyces virens and Streptomyces aurorectus.[6]
Biochemical relevance
[edit]The compound is capable of interrupting the synthesis of peptides.[7]
See also
[edit]References
[edit]- ^ Bartholomé, Axel; Janso, Jeffrey E; Reilly, Usa; O'Hagan, David (2017). "Fluorometabolite biosynthesis: isotopically labelled glycerol incorporations into the antibiotic nucleocidin in Streptomyces calvus". Organic & Biomolecular Chemistry. 15 (1): 61–64. doi:10.1039/c6ob02291j. hdl:10023/12031. PMID 27845468.
- ^ Pasternak, Ola; Zechel, David (May 2022). "Identification of Genes Essential for Sulfamate and Fluorine Incorporation During Nucleocidin Biosynthesis". The FASEB Journal. 36 (S1): fasebj.2022.36.S1.R2700. doi:10.1096/fasebj.2022.36.S1.R2700. hdl:1974/30179. ISSN 0892-6638. PMID 35552629. S2CID 248654450.
- ^ Carvalho, Maria F.; Oliveira, Rui S. (2017-10-03). "Natural production of fluorinated compounds and biotechnological prospects of the fluorinase enzyme". Critical Reviews in Biotechnology. 37 (7): 880–897. doi:10.1080/07388551.2016.1267109. hdl:10400.22/13870. ISSN 0738-8551. PMID 28049355. S2CID 27301409.
- ^ Shuman, Dennis A.; Robins, Morris J.; Robins, Roland K. (June 1970). "Synthesis of Nucleoside Sulfamates Related to Nucleocidin". Journal of the American Chemical Society. 92 (11): 3434–3440. doi:10.1021/ja00714a035. ISSN 0002-7863. PMID 5422764 – via JACS.
- ^ Carvalho, Maria F.; Oliveira, Rui S (October 2017). "Natural Production of Fluorinated Compounds and Biotechnological Prospects of the Fluorinate Enzyme". Critical Reviews in Biotechnology. 37 (7): 880–897. doi:10.1080/07388551.2016.1267109. hdl:10400.22/13870. ISSN 0738-8551. PMID 28049355. S2CID 27301409.
- ^ Pasternak, Ola; Zechel, David (May 2022). "Identification of Genes Essential for Sulfamate and Fluorine Incorporation During Nucleocidin Biosynthesis". The FASEB Journal. 36 (S1): fasebj.2022.36.S1.R2700. doi:10.1096/fasebj.2022.36.S1.R2700. hdl:1974/30179. ISSN 0892-6638. PMID 35552629. S2CID 248654450.
- ^ Bloch, A.; Coutsogeorgopoulos, C. (1971-11-01). "Inhibition of protein synthesis by 5'-sulfamoyladenosine". Biochemistry. 10 (24): 4394–4398. doi:10.1021/bi00800a007. ISSN 0006-2960. PMID 4946919.
