User:Mr. Ibrahem/Buspirone
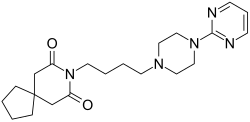 | |
| Clinical data | |
|---|---|
| Pronunciation | /ˈbjuːspɪroʊn/ (BEW-spi-rohn) |
| Trade names | Buspar, Namanspin |
| Other names | MJ 9022-1 |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a688005 |
| Pregnancy category |
|
| Routes of administration | By mouth |
| Legal status | |
| Legal status | |
| Pharmacokinetic data | |
| Bioavailability | 3.9%[1] |
| Protein binding | 86–95%[2] |
| Metabolism | Liver (via CYP3A4)[3][4] |
| Metabolites | 5-OH-Buspirone; 6-OH-Buspirone; 8-OH-Buspirone; 1-PP |
| Elimination half-life | 2.5 hours[3] |
| Excretion | Urine: 29–63%[2] Feces: 18–38%[2] |
| Identifiers | |
| |
| Chemical and physical data | |
| Formula | C21H31N5O2 |
| Molar mass | 385.512 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
| (verify) | |
Buspirone, sold under the brand name Buspar among others, is a medication primarily used to treat anxiety disorders, particularly generalized anxiety disorder.[5][6] Benefits support its short term use.[7] It has not been found to be effective in treating psychosis.[5] It is taken by mouth, and it may take up to four weeks for an effect.[5][6]
Common side effects include nausea, headaches, dizziness, and trouble concentrating.[5][7] Serious side effects may include hallucinations, serotonin syndrome, and seizures.[7] Use in pregnancy appears to be safe but has not been well studied, while use during breastfeeding is not recommended.[7][8] How it works is not clear but it is unrelated to benzodiazepines.[5]
Buspirone was first made in 1968 and approved for medical use in the United States in 1986.[5][6] It is available as a generic medication.[7] A month supply in the United Kingdom costs the NHS about £10 as of 2019.[7] In the United States the wholesale cost of this amount is about US$3.[9] In 2017, it was the 80th most commonly prescribed medication in the United States, with more than ten million prescriptions.[10][11]
References
[edit]- ^ Loane C, Politis M (June 2012). "Buspirone: what is it all about?". Brain Research. 1461: 111–8. doi:10.1016/j.brainres.2012.04.032. PMID 22608068.
- ^ a b c "buspirone (Rx) - BuSpar, Buspirex, more." Medscape Reference. WebMD. Archived from the original on 13 July 2019. Retrieved 14 November 2013.
- ^ a b Mahmood I, Sahajwalla C (April 1999). "Clinical pharmacokinetics and pharmacodynamics of buspirone, an anxiolytic drug". Clinical Pharmacokinetics. 36 (4): 277–87. doi:10.2165/00003088-199936040-00003. PMID 10320950. Archived (PDF) from the original on 2021-08-28. Retrieved 2019-09-06.
- ^ Zhu M, Zhao W, Jimenez H, Zhang D, Yeola S, Dai R, et al. (April 2005). "Cytochrome P450 3A-mediated metabolism of buspirone in human liver microsomes". Drug Metabolism and Disposition. 33 (4): 500–7. doi:10.1124/dmd.104.000836. PMID 15640381. S2CID 10142905.
- ^ a b c d e f g h "Buspirone Hydrochloride Monograph for Professionals". Drugs.com. American Society of Health-System Pharmacists. Archived from the original on 6 March 2019. Retrieved 3 March 2019.
- ^ a b c d Wilson, TK; Tripp, J (January 2018). "Buspirone". StatPearls. PMID 30285372. Archived from the original on 2020-08-11. Retrieved 2019-03-05.
- ^ a b c d e f g British national formulary : BNF 76 (76 ed.). Pharmaceutical Press. 2018. p. 338. ISBN 9780857113382.
- ^ "Buspirone Pregnancy and Breastfeeding Warnings". Drugs.com. Archived from the original on 6 March 2019. Retrieved 3 March 2019.
- ^ "NADAC as of 2019-02-27". Centers for Medicare and Medicaid Services. Archived from the original on 2019-03-06. Retrieved 3 March 2019.
- ^ "The Top 300 of 2020". ClinCalc. Archived from the original on 12 February 2021. Retrieved 11 April 2020.
- ^ "Buspirone - Drug Usage Statistics". ClinCalc. Archived from the original on 8 July 2020. Retrieved 11 April 2020.
