User:Mr. Ibrahem/Ganaxolone
 | |
| Clinical data | |
|---|---|
| Trade names | Ztalmy |
| Other names | GNX; CCD-1042; 3β-Methyl-5α-pregnan-3α-ol-20-one; 3α-Hydroxy-3β-methyl-5α-pregnan-20-one |
| AHFS/Drugs.com | Monograph |
| License data |
|
| Routes of administration | By mouth |
| Drug class | Neurosteroid |
| Legal status | |
| Legal status | |
| Identifiers | |
| |
| Chemical and physical data | |
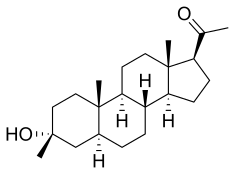
| Formula | C22H36O2 |
| Molar mass | 332.528 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
| | |
Ganaxolone, sold under the brand name Ztalmy, is a medication used to treat seizures in people with cyclin-dependent kinase-like 5 (CDKL5) deficiency disorder (CDD).[1] It may be used in those who are at least 2 years old.[1] It is taken by mouth.[1]
Common side effects include sleepiness, fever, increased saliva, and seasonal allergies.[1] Other side effects may include suicidal thoughts.[1] It is a neuroactive steroid gamma-aminobutyric acid (GABA) A receptor positive modulator.[1]
Ganaxolone was approved for medical use in the United States in 2022.[1] It is an orphan drug in Europe and not approved in the United Kingdom as of 2022.[2] In the United States it costs about 133,000 USD for a person who is around 4.5 years old as of 2022.[2]
References[edit]
- ^ a b c d e f g h i j "DailyMed - ZTALMY- ganaxolone suspension". dailymed.nlm.nih.gov. Archived from the original on 12 December 2022. Retrieved 11 December 2022.
- ^ a b "Ganaxolone". SPS - Specialist Pharmacy Service. 17 September 2020. Archived from the original on 25 September 2021. Retrieved 11 December 2022.
