User:Mr. Ibrahem/Ixazomib
 | |
| Clinical data | |
|---|---|
| Trade names | Ninlaro |
| Other names | Ixazomib citrate, MLN2238 |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a616008 |
| License data | |
| Pregnancy category |
|
| Routes of administration | By mouth (capsules) |
| Drug class | Proteasome inhibitor[1] |
| Legal status | |
| Legal status |
|
| Pharmacokinetic data | |
| Bioavailability | 58%[2] |
| Protein binding | 99% |
| Metabolism | Hepatic (CYP: 3A4 (42%), 1A2 (26%), 2B6 (16%) and others) |
| Elimination half-life | 9.5 days |
| Excretion | Urine (62%), faeces (22%) |
| Identifiers | |
| |
| Chemical and physical data | |
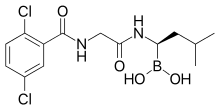
| Formula | C14H19BCl2N2O4 |
| Molar mass | 361.03 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
Ixazomib, sold under the brand name Ninlaro, is a medication used to treat multiple myeloma.[1] It is used together with lenalidomide and dexamethasone in people who have failed other treatment.[1] It is taken by mouth.[3]
Common side effects include diarrhea, constipation, low platelets, low white blood cells, peripheral neuropathy, nausea, and swelling.[1] Other side effects may include liver problems.[3] Use in pregnancy may harm the baby.[3] It is a proteasome inhibitor and works by preventing protein breakdown in cells.[1]
Ixazomib was approved for medical use in the United States in 2015 and Europe in 2016.[3][1] In the United Kingdom 4 weeks of treatment costs the NHS about £6,300 as of 2021.[4] In the United States this amount costs about 11,400 USD.[5]
References[edit]
- ^ a b c d e f g h "Ninlaro". European Medicines Agency. Archived from the original on 14 November 2021. Retrieved 1 December 2021.
- ^ "Ninlaro- ixazomib capsule". DailyMed. 24 March 2020. Archived from the original on 27 October 2020. Retrieved 19 August 2020.
- ^ a b c d "Ixazomib Citrate Monograph for Professionals". Drugs.com. Archived from the original on 13 May 2021. Retrieved 26 November 2021.
- ^ BNF 81: March-September 2021. BMJ Group and the Pharmaceutical Press. 2021. p. 1009. ISBN 978-0857114105.
- ^ "Ninlaro Prices, Coupons & Patient Assistance Programs". Drugs.com. Archived from the original on 21 August 2021. Retrieved 1 December 2021.
