User:Mr. Ibrahem/Uridine triacetate
 | |
| Clinical data | |
|---|---|
| Trade names | Vistogard, Xuriden |
| Other names | Vistonuridine |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a616020 |
| License data | |
| Routes of administration | By mouth |
| Legal status | |
| Legal status |
|
| Pharmacokinetic data | |
| Metabolism | Pyrimidine catabolic pathway |
| Onset of action | Tmax = 2–3 hours |
| Elimination half-life | 2–2.5 hours |
| Excretion | Kidney |
| Identifiers | |
| |
| Chemical and physical data | |
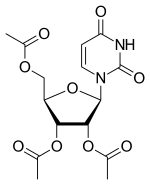
| Formula | C15H18N2O9 |
| Molar mass | 370.314 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
Uridine triacetate, formerly known as vistonuridine, is a medication used to treat hereditary orotic aciduria and poisoning due to fluorouracil or capecitabine.[1] For poisoning it should be used within 4 days and in an overdose may be used regardless of the presence of symptoms.[1] It is taken by mouth.[1][2]
Side effects may include nausea or diarrhea.[1] Use in pregnancy and breastfeeding is of unclear safety.[3] It is a prodrug of uridine; by which it acts.[2][1]
Uridine triacetate was approved for medical use in the United States in 2015.[1] It is not approved in Europe.[4] In the United States it costs about 4,200 USD per 10 gram dose as of 2021.[5] In the United States it is only available through specialty pharmacies.[1] In Canada approval is required from the Health Canada Special Access Programme.[6] Brand names include Vistogard and Xuriden.[1]
References[edit]
- ^ a b c d e f g h i "Uridine Triacetate Monograph for Professionals". Drugs.com. Retrieved 13 September 2021.
- ^ a b Saif, Muhammad Wasif (4 March 2019). "Uridine triacetate - an antidote in the treatment of 5-fluorouracil or capecitabine poisoning". Expert Opinion on Orphan Drugs. 7 (3): 95–103. doi:10.1080/21678707.2019.1591273.
- ^ "Uridine Use During Pregnancy". Drugs.com. Retrieved 13 September 2021.
- ^ "FDA approves drug for genetic condition that affects 20 patients worldwide". The Pharmaceutical Journal. 2015. doi:10.1211/PJ.2015.20069307.
- ^ "Vistogard Prices, Coupons & Patient Assistance Programs". Drugs.com. Retrieved 13 September 2021.
- ^ "BC Cancer Agency Management Guidelines Management of 5-fluorouracil (5FU) infusion overdose" (PDF). Retrieved 13 September 2021.
