User:SalFromOuterSpace/sandbox
Default mode network[edit]
| Default mode network | |
|---|---|
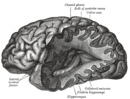
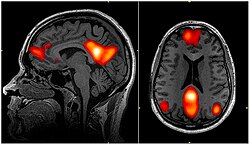 fMRI scan showing regions of the default mode network; the dorsal medial prefrontal cortex, the posterior cingulate cortex, the precuneus and the angular gyrus | |
| Anatomical terminology |
In neuroscience, the default mode network (DMN), also known as the default network, default state network, or anatomically the medial frontoparietal network (M-FPN), is a large-scale brain network primarily composed of the dorsal medial prefrontal cortex, posterior cingulate cortex, precuneus and angular gyrus. It is best known for being active when a person is not focused on the outside world and the brain is at wakeful rest, such as during daydreaming and mind-wandering. It can also be active during detailed thoughts related to external task performance.[3] Other times that the DMN is active include when the individual is thinking about others, thinking about themselves, remembering the past, and planning for the future.[4][5]
The DMN was originally noticed to be deactivated in certain goal-oriented tasks and was sometimes referred to as the task-negative network,[6] in contrast with the task-positive network. This nomenclature is now widely considered misleading, because the network can be active in internal goal-oriented and conceptual cognitive tasks.[7][8][9][10] The DMN has been shown to be negatively correlated with other networks in the brain such as attention networks.[11]
Evidence has pointed to disruptions in the DMN of people with Alzheimer's disease and autism spectrum disorder.[4]
History[edit]
Hans Berger, the inventor of the electroencephalogram, was the first to propose the idea that the brain is constantly busy. In a series of papers published in 1929, he showed that the electrical oscillations detected by his device do not cease even when the subject is at rest. However, his ideas were not taken seriously, and a general perception formed among neurologists that only when a focused activity is performed does the brain (or a part of the brain) become active.[12]
But in the 1950s, Louis Sokoloff and his colleagues noticed that metabolism in the brain stayed the same when a person went from a resting state to performing effortful math problems, suggesting active metabolism in the brain must also be happening during rest.[4] In the 1970s, David H. Ingvar and colleagues observed blood flow in the front part of the brain became the highest when a person is at rest.[4] Around the same time, intrinsic oscillatory behavior in vertebrate neurons was observed in cerebellar Purkinje cells, inferior olivary nucleus and thalamus.[13]
In the 1990s, with the advent of positron emission tomography (PET) scans, researchers began to notice that when a person is involved in perception, language, and attention tasks, the same brain areas become less active compared to passive rest, and labeled these areas as becoming "deactivated".[4]
In 1995, Bharat Biswal, a graduate student at the Medical College of Wisconsin in Milwaukee, discovered that the human sensorimotor system displayed "resting-state connectivity," exhibiting synchronicity in functional magnetic resonance imaging (fMRI) scans while not engaged in any task.[14][15]
Later, experiments by neurologist Marcus E. Raichle's lab at Washington University School of Medicine and other groups[16] showed that the brain's energy consumption is increased by less than 5% of its baseline energy consumption while performing a focused mental task. These experiments showed that the brain is constantly active with a high level of activity even when the person is not engaged in focused mental work. Research thereafter focused on finding the regions responsible for this constant background activity level.[12]
Raichle coined the term "default mode" in 2001 to describe resting state brain function;[17] the concept rapidly became a central theme in neuroscience.[18] Around this time the idea was developed that this network of brain areas is involved in internally directed thoughts and is suspended during specific goal-directed behaviors. In 2003, Greicius and colleagues examined resting state fMRI scans and looked at how correlated different sections in the brain are to each other. Their correlation maps highlighted the same areas already identified by the other researchers.[19] This was important because it demonstrated a convergence of methods all leading to the same areas being involved in the DMN. Since then other networks have been identified, such as visual, auditory, and attention networks. Some of them are often anti-correlated with the default mode network.[11]
Until the mid-2000s, researchers labeled the default mode network as the "task-negative network" because it was deactivated when participants had to perform external goal-directed tasks.[6] DMN was thought to only be active during passive rest and inactive during tasks. However, more recent studies have demonstrated the DMN to be active in certain internal goal-directed tasks such as social working memory and autobiographical tasks.[7]
Around 2007, the number of papers referencing the default mode network skyrocketed.[20] In all years prior to 2007, there were 12 papers published that referenced "default mode network" or "default network" in the title; however, between 2007 and 2014 the number increased to 1,384 papers. One reason for the increase in papers was the robust effect of finding the DMN with resting-state scans and independent component analysis (ICA).[16][21] Another reason was that the DMN could be measured with short and effortless resting-state scans, meaning they could be performed on any population including young children, clinical populations, and nonhuman primates.[4] A third reason was that the role of the DMN had been expanded to more than just a passive brain network.[4]
Anatomy[edit]
This article needs attention from an expert in neuroscience or anatomy. See the talk page for details. (February 2023) |

The default mode network is an interconnected and anatomically defined[4] set of brain regions. The network can be separated into hubs and subsections:
Functional hubs:[23] Information regarding the self
- Posterior cingulate cortex (PCC) & precuneus: Combines bottom-up (not controlled) attention with information from memory and perception. The ventral (lower) part of PCC activates in all tasks which involve the DMN including those related to the self, related to others, remembering the past, thinking about the future, and processing concepts plus spatial navigation. The dorsal (upper) part of PCC involves involuntary awareness and arousal. The precuneus is involved in visual, sensorimotor, and attentional information.
- Medial prefrontal cortex (mPFC): Decisions about self-processing such as personal information, autobiographical memories, future goals and events, and decision making regarding those personally very close such as family. The ventral (lower) part is involved in positive emotional information and internally valued reward.
- Angular gyrus: Connects perception, attention, spatial cognition, and action and helps with parts of recall of episodic memories.
Dorsal medial subsystem:[23] Thinking about others
- Functional hubs: PCC, mPFC, and angular gyrus
- Dorsal medial prefrontal cortex (dmPFC): Involved in social directed thought such as determining or inferring the purpose of others' actions
- Temporoparietal junction (TPJ): Reflects on beliefs about others, also known as theory of mind
- Lateral temporal cortex: Retrieval of social semantic and conceptual knowledge
- Anterior temporal pole: Abstract conceptual information particularly social in nature
Medial temporal subsystem:[23] Autobiographical memory and future simulations
- Functional hubs: PCC, mPFC, and angular gyrus
- Hippocampus (HF+): Formation of new memories as well as remembering the past and imagining the future
- Parahippocampus (PHC): Spatial and scene recognition and simulation
- Retrosplenial cortex (RSC): Spatial navigation[24]
- Posterior inferior parietal lobe (pIPL): Junction of auditory, visual, and somatosensory information and attention
The default mode network is most commonly defined with resting state data by putting a seed in the posterior cingulate cortex and examining which other brain areas most correlate with this area.[19] The DMN can also be defined by the areas deactivated during external directed tasks compared to rest.[17] Independent component analysis (ICA) robustly finds the DMN for individuals and across groups, and has become the standard tool for mapping the default network.[16][21]
It has been shown that the default mode network exhibits the highest overlap in its structural and functional connectivity, which suggests that the structural architecture of the brain may be built in such a way that this particular network is activated by default.[1] Recent evidence from a population brain-imaging study of 10,000 UK Biobank participants further suggests that each DMN node can be decomposed into subregions with complementary structural and functional properties. It has been a widespread practice in DMN research to treat its constituent nodes to be functionally homogeneous, but the distinction between subnodes within each major DMN node has mostly been neglected. However, the close proximity of subnodes that propagate hippocampal space-time outputs and subnodes that describe the global network architecture may enable default functions, such as autobiographical recall or internally-orientated thinking.[25]
In the infant's brain, there is limited evidence of the default network, but default network connectivity is more consistent in children aged 9–12 years, suggesting that the default network undergoes developmental change.[11]
Functional connectivity analysis in monkeys shows a similar network of regions to the default mode network seen in humans.[4] The PCC is also a key hub in monkeys; however, the mPFC is smaller and less well connected to other brain regions, largely because human's mPFC is much larger and well developed.[4]
Diffusion MRI imaging shows white matter tracts connecting different areas of the DMN together.[20] The structural connections found from diffusion MRI imaging and the functional correlations from resting state fMRI show the highest level of overlap and agreement within the DMN areas.[1] This provides evidence that neurons in the DMN regions are linked to each other through large tracts of axons and this causes activity in these areas to be correlated with one another. From the point of view of effective connectivity, many studies have attempted to shed some light using dynamic causal modeling, with inconsistent results. However, directionality from the medial prefrontal cortex towards the posterior cingulate gyrus seems confirmed in multiple studies, and the inconsistent results appear to be related to small sample size analysis.[26]
Function[edit]
The default mode network is thought to be involved in several different functions:
It is potentially the neurological basis for the self:[20]
- Autobiographical information: Memories of collection of events and facts about one's self
- Self-reference: Referring to traits and descriptions of one's self
- Emotion of one's self: Reflecting about one's own emotional state
Thinking about others:[20]
- Theory of mind: Thinking about the thoughts of others and what they might or might not know
- Emotions of others: Understanding the emotions of other people and empathizing with their feelings
- Moral reasoning: Determining a just and an unjust result of an action
- Social evaluations: Good-bad attitude judgements about social concepts
- Social categories: Reflecting on important social characteristics and status of a group
- Social isolation: A perceived lack of social interaction[27]
Remembering the past and thinking about the future:[20]
- Remembering the past: Recalling events that happened in the past
- Imagining the future: Envisioning events that might happen in the future
- Episodic memory: Detailed memory related to specific events in time
- Story comprehension: Understanding and remembering a narrative
- Replay: Consolidating recently acquired memory traces[28]
The default mode network is active during passive rest and mind-wandering[4] which usually involves thinking about others, thinking about one's self, remembering the past, and envisioning the future rather than the task being performed.[20] Recent work, however, has challenged a specific mapping between the default mode network and mind-wandering, given that the system is important in maintaining detailed representations of task information during working memory encoding.[29] Electrocorticography studies (which involve placing electrodes on the surface of a subject's cerebral cortex) have shown the default mode network becomes activated within a fraction of a second after participants finish a task.[30] Additionally, during attention demanding tasks, sufficient deactivation of the default mode network at the time of memory encoding has been shown to result in more successful long-term memory consolidation.[31]
Studies have shown that when people watch a movie,[32] listen to a story,[33][34] or read a story,[35] their DMNs are highly correlated with each other. DMNs are not correlated if the stories are scrambled or are in a language the person does not understand, suggesting that the network is highly involved in the comprehension and the subsequent memory formation of that story.[34] The DMN is shown to even be correlated if the same story is presented to different people in different languages,[36] further suggesting the DMN is truly involved in the comprehension aspect of the story and not the auditory or language aspect.
The default mode network is deactivated during some external goal-oriented tasks such as visual attention or cognitive working memory tasks.[6] However, with internal goal-oriented tasks, such as social working memory or autobiographical tasks, the DMN is positively activated with the task and correlates with other networks such as the network involved in executive function.[7] Regions of the DMN are also activated during cognitively demanding tasks that require higher-order conceptual representations.[9] The DMN shows higher activation when behavioral responses are stable, and this activation is independent of self-reported mind wandering.[37]
Tsoukalas (2017) links theory of mind to immobilization, and suggests that the default network is activated by the immobilization inherent in the testing procedure (the patient is strapped supine on a stretcher and inserted by a narrow tunnel into a massive metallic structure). This procedure creates a sense of entrapment and, not surprisingly, the most commonly reported side-effect is claustrophobia.[38]
Gabrielle et al. (2019) suggests that the DMN is related to the perception of beauty, in which the network becomes activated in a generalized way to aesthetically moving domains such as artworks, landscapes, and architecture. This would explain a deep inner feeling of pleasure related to aesthetics, interconnected with the sense of personal identity, due to the network functions related to the self.[39]
Clinical significance[edit]
The default mode network has been hypothesized to be relevant to disorders including Alzheimer's disease, autism, schizophrenia, major depressive disorder (MDD), chronic pain, post-traumatic stress disorder (PTSD) and others.[4][40] In particular, the DMN has also been reported to show overlapping yet distinct neural activity patterns across different mental health conditions, such as when directly comparing attention deficit hyperactivity disorder (ADHD) and autism.[41]
People with Alzheimer's disease show a reduction in glucose (energy use) within the areas of the default mode network.[4] These reductions start off as slight decreases in patients with mild symptoms and continue to large reductions in those with severe symptoms. Surprisingly, disruptions in the DMN begin even before individuals show signs of Alzheimer's disease.[4] Plots of the peptide amyloid-beta, which is thought to cause Alzheimer's disease, show the buildup of the peptide is within the DMN.[4] This prompted Randy Buckner and colleagues to propose the high metabolic rate from continuous activation of DMN causes more amyloid-beta peptide to accumulate in these DMN areas.[4] These amyloid-beta peptides disrupt the DMN and because the DMN is heavily involved in memory formation and retrieval, this disruption leads to the symptoms of Alzheimer's disease.
DMN is thought to be disrupted in individuals with autism spectrum disorder.[4][42] These individuals are impaired in social interaction and communication which are tasks central to this network. Studies have shown worse connections between areas of the DMN in individuals with autism, especially between the mPFC (involved in thinking about the self and others) and the PCC (the central core of the DMN).[43][44] The more severe the autism, the less connected these areas are to each other.[43][44] It is not clear if this is a cause or a result of autism, or if a third factor is causing both (confounding).
Although it is not clear whether the DMN connectivity is increased or decreased in psychotic bipolar disorder and schizophrenia, several genes correlated with altered DMN connectivity are also risk genes for mood and psychosis disorders.[45]
Rumination, one of the main symptoms of major depressive disorder, is associated with increased DMN connectivity and dominance over other networks during rest.[46][47] Such DMN hyperconnectivity has been observed in first-episode depression[48] and chronic pain.[49] Altered DMN connectivity may change the way a person perceives events and their social and moral reasoning, thus increasing their susceptibility to depressive symptoms.[50]
Lower connectivity between brain regions was found across the default network in people who have experienced long-term trauma, such as childhood abuse or neglect, and is associated with dysfunctional attachment patterns. Among people experiencing PTSD, lower activation was found in the posterior cingulate gyrus compared to controls, and severe PTSD was characterized by lower connectivity within the DMN.[40][51]
Adults and children with ADHD show reduced anticorrelation between the DMN and other brain networks.[52][53] The cause may be a lag in brain maturation.[54] More generally, competing activation between the DMN and other networks during memory encoding may result in poor long-term memory consolidation, which is a symptom of not only ADHD but also depression, anxiety, autism, and schizophrenia.[31]
Modulation[edit]
The default mode network (DMN) may be modulated by the following interventions and processes:
- Acupuncture – Deactivation of the limbic brain areas and the DMN.[55] It has been suggested that this is due to the pain response.[56]
- Antidepressants – Abnormalities in DMN connectivity are reduced following treatment with antidepressant medications in PTSD.[57]
- Attention Training Technique - Research shows that even a single session of Attention Training Technique changes functional connectivity of the DMN.[58]
- Deep brain stimulation – Alterations in brain activity with deep brain stimulation may be used to balance resting state networks.[59]
- Meditation – Structural changes in areas of the DMN such as the temporoparietal junction, posterior cingulate cortex, and precuneus have been found in meditation practitioners.[60] There is reduced activation and reduced functional connectivity of the DMN in long-term practitioners.[60] Various forms of nondirective meditation, including Transcendental Meditation[61] and Acem Meditation,[62] have been found to activate the DMN.
- Physical Activity and Exercise – Physical Activity, and more likely Aerobic Training, may alter the DMN. In addition, sports experts are showing networks differences, notably of the DMN.[63][64][65]
- Psychedelic drugs – Reduced blood flow to the PCC and mPFC was observed under the administration of psilocybin. These two areas are considered to be the main nodes of the DMN.[66] One study on the effects of LSD demonstrated that the drug desynchronizes brain activity within the DMN; the activity of the brain regions that constitute the DMN becomes less correlated.[67]
- Psychotherapy – In PTSD, the abnormalities in the default mode network normalize in individuals who respond to psychotherapy interventions.[68][57]
- Sleep deprivation – Functional connectivity between nodes of the DMN in their resting-state is usually strong, but sleep deprivation results in a decrease in connectivity within the DMN.[69] Recent studies suggest a decrease in connectivity between the DMN and the task-positive network as a result of sleep loss.[70]
- Sleeping and resting wakefulness
- Onset of sleep – Increase in connectivity between the DMN and the task-positive network.[71]
- REM sleep – Possible increase in connectivity between nodes of the DMN.[71]
- Resting wakefulness – Functional connectivity between nodes of the DMN is strong.[71]
- Stage N2 of NREM sleep – Decrease in connectivity between the posterior cingulate cortex and medial prefrontal cortex.[71]
- Stage N3 of NREM sleep – Further decrease in connectivity between the PCC and MPFC.[71]
Criticism[edit]
Some have argued the brain areas in the default mode network only show up together because of the vascular coupling of large arteries and veins in the brain near these areas, not because these areas are actually functionally connected to each other. Support for this argument comes from studies that show changing in breathing alters oxygen levels in the blood which in turn affects DMN the most.[4] These studies however do not explain why the DMN can also be identified using PET scans by measuring glucose metabolism which is independent of vascular coupling[4] and in electrocorticography studies[72] measuring electrical activity on the surface of the brain, and in MEG by measuring magnetic fields associated with electrophysiological brain activity that bypasses the hemodynamic response.[73]
The idea of a "default network" is not universally accepted.[74] In 2007 the concept of the default mode was criticized as not being useful for understanding brain function, on the grounds that a simpler hypothesis is that a resting brain actually does more processing than a brain doing certain "demanding" tasks, and that there is no special significance to the intrinsic activity of the resting brain.[75]
Nomenclature[edit]
The default mode network has also been called the language network, semantic system, or limbic network.[10] Even though the dichotomy is misleading,[7] the term task-negative network is still sometimes used to contrast it against other more externally-oriented brain networks.[53]
In 2019, Uddin et al. proposed that medial frontoparietal network (M-FPN) be used as a standard anatomical name for this network.[10]
See also[edit]
References[edit]
- ^ a b c Horn, Andreas; Ostwald, Dirk; Reisert, Marco; Blankenburg, Felix (2013). "The structural-functional connectome and the default mode network of the human brain". NeuroImage. 102: 142–151. doi:10.1016/j.neuroimage.2013.09.069. PMID 24099851. S2CID 6455982.
- ^ Garrity, A.; Pearlson, G. D.; McKiernan, K.; Lloyd, D.; Kiehl, K. A.; Calhoun, V. D. (2007). "Aberrant default mode functional connectivity in schizophrenia". Am. J. Psychiatry. 164 (3): 450–457. doi:10.1176/ajp.2007.164.3.450. PMID 17329470.
- ^ Sormaz, Mladen; Murphy, Charlotte; Wang, Hao-Ting; Hymers, Mark; Karapanagiotidis, Theodoros; Poerio, Giulia; Margulies, Daniel S.; Jefferies, Elizabeth; Smallwood, Jonathan (2018). "Default mode network can support the level of detail in experience during active task states". Proceedings of the National Academy of Sciences. 115 (37): 9318–9323. Bibcode:2018PNAS..115.9318S. doi:10.1073/pnas.1721259115. PMC 6140531. PMID 30150393.
- ^ a b c d e f g h i j k l m n o p q r s Buckner, R. L.; Andrews-Hanna, J. R.; Schacter, D. L. (2008). "The Brain's Default Network: Anatomy, Function, and Relevance to Disease". Annals of the New York Academy of Sciences. 1124 (1): 1–38. Bibcode:2008NYASA1124....1B. CiteSeerX 10.1.1.689.6903. doi:10.1196/annals.1440.011. PMID 18400922. S2CID 3167595.
- ^ Lieberman, Matthew (2 September 2016). Social. Broadway Books. p. 19. ISBN 978-0-307-88910-2.
- ^ a b c Fox, Michael D.; Snyder, Abraham Z.; Vincent, Justin L.; Corbetta, Maurizio; Van Essen, David C.; Raichle, Marcus E. (5 July 2005). "The human brain is intrinsically organized into dynamic, anticorrelated functional networks". Proceedings of the National Academy of Sciences of the United States of America. 102 (27): 9673–9678. Bibcode:2005PNAS..102.9673F. doi:10.1073/pnas.0504136102. ISSN 0027-8424. PMC 1157105. PMID 15976020.
- ^ a b c d Spreng, R. Nathan (1 January 2012). "The fallacy of a "task-negative" network". Frontiers in Psychology. 3: 145. doi:10.3389/fpsyg.2012.00145. ISSN 1664-1078. PMC 3349953. PMID 22593750.
- ^ Fortenbaugh, FC; DeGutis, J; Esterman, M (May 2017). "Recent theoretical, neural, and clinical advances in sustained attention research". Annals of the New York Academy of Sciences. 1396 (1): 70–91. Bibcode:2017NYASA1396...70F. doi:10.1111/nyas.13318. PMC 5522184. PMID 28260249.
- ^ a b Murphy, C; Jefferies, E; Rueschemeyer, SA; Sormaz, M; Wang, HT; Margulies, DS; Smallwood, J (1 May 2018). "Distant from input: Evidence of regions within the default mode network supporting perceptually-decoupled and conceptually-guided cognition". NeuroImage. 171: 393–401. doi:10.1016/j.neuroimage.2018.01.017. PMC 5883322. PMID 29339310.
- ^ a b c Uddin, Lucina Q.; Yeo, B. T. Thomas; Spreng, R. Nathan (1 November 2019). "Towards a Universal Taxonomy of Macro-scale Functional Human Brain Networks". Brain Topography. 32 (6): 926–942. doi:10.1007/s10548-019-00744-6. ISSN 1573-6792. PMC 7325607. PMID 31707621.
- ^ a b c Broyd, Samantha J.; Demanuele, Charmaine; Debener, Stefan; Helps, Suzannah K.; James, Christopher J.; Sonuga-Barke, Edmund J. S. (2009). "Default-mode brain dysfunction in mental disorders: A systematic review". Neuroscience & Biobehavioral Reviews. 33 (3): 279–96. doi:10.1016/j.neubiorev.2008.09.002. PMID 18824195. S2CID 7175805.
- ^ a b Raichle, Marcus (March 2010). "The Brain's Dark Energy". Scientific American. 302 (3): 44–49. Bibcode:2010SciAm.302c..44R. doi:10.1038/scientificamerican0310-44. PMID 20184182.
- ^ Llinas, R. R. (2014). "Intrinsic electrical properties of mammalian neurons and CNS function: a historical perspective". Front Cell Neurosci. 8: 320. doi:10.3389/fncel.2014.00320. PMC 4219458. PMID 25408634.
- ^ Biswal, B; Yetkin, F. Z.; Haughton, V. M.; Hyde, J. S. (1995). "Functional connectivity in the motor cortex of resting human brain using echoplanar MRI". Magn Reson Med. 34 (4): 537–541. doi:10.1002/mrm.1910340409. PMID 8524021. S2CID 775793.
- ^ Shen, H. H. (2015). "Core Concepts: Resting State Connectivity". Proceedings of the National Academy of Sciences. 112 (46): 14115–14116. Bibcode:2015PNAS..11214115S. doi:10.1073/pnas.1518785112. PMC 4655520. PMID 26578753.
- ^ a b c Kiviniemi, Vesa J.; Kantola, Juha-Heikki; Jauhiainen, Jukka; Hyvärinen, Aapo; Tervonen, Osmo (2003). "Independent component analysis of nondeterministic fMRI signal sources". NeuroImage. 19 (2 Pt 1): 253–260. doi:10.1016/S1053-8119(03)00097-1. PMID 12814576. S2CID 17110486.
- ^ a b Raichle, M. E.; MacLeod, A. M.; Snyder, A. Z.; Powers, W. J.; Gusnard, D. A.; Shulman, G. L. (2001). "Inaugural Article: A default mode of brain function". Proceedings of the National Academy of Sciences. 98 (2): 676–82. Bibcode:2001PNAS...98..676R. doi:10.1073/pnas.98.2.676. PMC 14647. PMID 11209064.
- ^ Raichle, Marcus E.; Snyder, Abraham Z. (2007). "A default mode of brain function: A brief history of an evolving idea". NeuroImage. 37 (4): 1083–90. doi:10.1016/j.neuroimage.2007.02.041. PMID 17719799. S2CID 3380973.
- ^ a b Greicius, Michael D.; Krasnow, Ben; Reiss, Allan L.; Menon, Vinod (7 January 2003). "Functional connectivity in the resting brain: a network analysis of the default mode hypothesis". Proceedings of the National Academy of Sciences of the United States of America. 100 (1): 253–258. Bibcode:2003PNAS..100..253G. doi:10.1073/pnas.0135058100. ISSN 0027-8424. PMC 140943. PMID 12506194.
- ^ a b c d e f Andrews-Hanna, Jessica R. (1 June 2012). "The brain's default network and its adaptive role in internal mentation". The Neuroscientist. 18 (3): 251–270. doi:10.1177/1073858411403316. ISSN 1089-4098. PMC 3553600. PMID 21677128.
- ^ a b De Luca, M; Beckmann, CF; De Stefano, N; Matthews, PM; Smith, SM (15 February 2006). "fMRI resting state networks define distinct modes of long-distance interactions in the human brain". NeuroImage. 29 (4): 1359–1367. doi:10.1016/j.neuroimage.2005.08.035. PMID 16260155. S2CID 16193549.
- ^ Fair, Damien A.; Cohen, Alexander L.; Power, Jonathan D.; Dosenbach, Nico U. F.; Church, Jessica A.; Miezin, Francis M.; Schlaggar, Bradley L.; Petersen, Steven E. (2009). Sporns, Olaf (ed.). "Functional Brain Networks Develop from a 'Local to Distributed' Organization". PLOS Computational Biology. 5 (5): e1000381. Bibcode:2009PLSCB...5E0381F. doi:10.1371/journal.pcbi.1000381. PMC 2671306. PMID 19412534.
- ^ a b c Andrews-Hanna, Jessica R.; Smallwood, Jonathan; Spreng, R. Nathan (1 May 2014). "The default network and self-generated thought: component processes, dynamic control, and clinical relevance". Annals of the New York Academy of Sciences. 1316 (1): 29–52. Bibcode:2014NYASA1316...29A. doi:10.1111/nyas.12360. ISSN 1749-6632. PMC 4039623. PMID 24502540.
- ^ Roseman, Moshe; Elias, Uri; Kletenik, Isaiah; Ferguson, Michael A.; Fox, Michael D.; Horowitz, Zalman; Marshall, Gad A.; Spiers, Hugo J.; Arzy, Shahar (2023-12-13). "A neural circuit for spatial orientation derived from brain lesions". Cerebral Cortex. 34 (1): bhad486. doi:10.1093/cercor/bhad486. ISSN 1460-2199. PMC 10793567. PMID 38100330.
- ^ Kernbach, J.M.; Yeo, B.T.T.; Smallwood, J.; Margulies, D.S.; Thiebaut; de Schotten, M.; Walter, H.; Sabuncu, M.R.; Holmes, A.J.; Gramfort, A.; Varoquaux, G.; Thirion, B.; Bzdok, D. (2018). "Subspecialization within default mode nodes characterized in 10,000 UK Biobank participants". Proc. Natl. Acad. Sci. 115 (48): 12295–12300. Bibcode:2018PNAS..11512295K. doi:10.1073/pnas.1804876115. PMC 6275484. PMID 30420501.
- ^ Silchenko, Alexander N.; Hoffstaedter, Felix; Eickhoff, Simon B. (2023). "Impact of sample size and regression of tissue-specific signals on effective connectivity within the core default mode network". Human Brain Mapping.
- ^ Spreng, R.N., Dimas, E., Mwilambwe-Tshilobo, L., Dagher, A., Koellinger, P., Nave, G., Ong, A., Kernbach, J.M., Wiecki, T.V., Ge, T., Holmes, A.J., Yeo, B.T.T., Turner, G.R., Dunbar, R.I.M., Bzdok, D (2020). "The default network of the human brain is associated with perceived social isolation". Nature Communications. 11 (1). Nature Publishing Group: 6393. Bibcode:2020NatCo..11.6393S. doi:10.1038/s41467-020-20039-w. PMC 7738683. PMID 33319780.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Higgins C, Liu Y, Vidaurre D, Kurth-Nelson Z, Dolan R, Behrens T, Woolrich M (March 2021). "Replay bursts in humans coincide with activation of the default mode and parietal alpha networks". Neuron. 109 (5): 882–893. doi:10.1016/j.neuron.2020.12.007. PMC 7927915. PMID 33357412.
- ^ Sormaz, Mladen; Murphy, Charlotte; Wang, Hao-ting; Hymers, Mark; Karapanagiotidis, Theodoros; Poerio, Giulia; Margulies, Daniel S.; Jefferies, Elizabeth; Smallwood, Jonathan (24 August 2018). "Default mode network can support the level of detail in experience during active task states". Proceedings of the National Academy of Sciences. 115 (37): 9318–9323. Bibcode:2018PNAS..115.9318S. doi:10.1073/pnas.1721259115. ISSN 0027-8424. PMC 6140531. PMID 30150393.
- ^ Dastjerdi, Mohammad; Foster, Brett L.; Nasrullah, Sharmin; Rauschecker, Andreas M.; Dougherty, Robert F.; Townsend, Jennifer D.; Chang, Catie; Greicius, Michael D.; Menon, Vinod (15 February 2011). "Differential electrophysiological response during rest, self-referential, and non-self-referential tasks in human posteromedial cortex". Proceedings of the National Academy of Sciences of the United States of America. 108 (7): 3023–3028. Bibcode:2011PNAS..108.3023D. doi:10.1073/pnas.1017098108. ISSN 1091-6490. PMC 3041085. PMID 21282630.
- ^ a b Lefebvre, Etienne; D’Angiulli, Amedeo (2019). "Imagery-Mediated Verbal Learning Depends on Vividness–Familiarity Interactions: The Possible Role of Dualistic Resting State Network Activity Interference". Brain Sciences. 9 (6): 143. doi:10.3390/brainsci9060143. ISSN 2076-3425. PMC 6627679. PMID 31216699.
- ^ Hasson, Uri; Furman, Orit; Clark, Dav; Dudai, Yadin; Davachi, Lila (7 February 2008). "Enhanced intersubject correlations during movie viewing correlate with successful episodic encoding". Neuron. 57 (3): 452–462. doi:10.1016/j.neuron.2007.12.009. ISSN 0896-6273. PMC 2789242. PMID 18255037.
- ^ Lerner, Yulia; Honey, Christopher J.; Silbert, Lauren J.; Hasson, Uri (23 February 2011). "Topographic mapping of a hierarchy of temporal receptive windows using a narrated story". The Journal of Neuroscience. 31 (8): 2906–2915. doi:10.1523/JNEUROSCI.3684-10.2011. ISSN 1529-2401. PMC 3089381. PMID 21414912.
- ^ a b Simony, Erez; Honey, Christopher J; Chen, Janice; Lositsky, Olga; Yeshurun, Yaara; Wiesel, Ami; Hasson, Uri (18 July 2016). "Dynamic reconfiguration of the default mode network during narrative comprehension". Nature Communications. 7 (1): 12141. Bibcode:2016NatCo...712141S. doi:10.1038/ncomms12141. ISSN 2041-1723. PMC 4960303. PMID 27424918.
- ^ Regev, Mor; Honey, Christopher J.; Simony, Erez; Hasson, Uri (2 October 2013). "Selective and invariant neural responses to spoken and written narratives". The Journal of Neuroscience. 33 (40): 15978–15988. doi:10.1523/JNEUROSCI.1580-13.2013. ISSN 1529-2401. PMC 3787506. PMID 24089502.
- ^ Honey, Christopher J.; Thompson, Christopher R.; Lerner, Yulia; Hasson, Uri (31 October 2012). "Not lost in translation: neural responses shared across languages". The Journal of Neuroscience. 32 (44): 15277–15283. doi:10.1523/JNEUROSCI.1800-12.2012. ISSN 1529-2401. PMC 3525075. PMID 23115166.
- ^ Kucyi, Aaron (2016). "Spontaneous default network activity reflects behavioral variability independent of mind-wandering". PNAS. 113 (48): 13899–13904. Bibcode:2016PNAS..11313899K. doi:10.1073/pnas.1611743113. PMC 5137714. PMID 27856733.
- ^ Tsoukalas, Ioannis (2017). "Theory of Mind: Towards an Evolutionary Theory". Evolutionary Psychological Science. 4: 38–66. doi:10.1007/s40806-017-0112-x.Pdf.
- ^ Starr, G. Gabrielle; Stahl, Jonathan L.; Belfi, Amy M.; Isik, Ayse Ilkay; Vessel, Edward A. (4 September 2019). "The default-mode network represents aesthetic appeal that generalizes across visual domains". Proceedings of the National Academy of Sciences. 116 (38): 19155–19164. Bibcode:2019PNAS..11619155V. doi:10.1073/pnas.1902650116. ISSN 0027-8424. PMC 6754616. PMID 31484756.
- ^ a b Akiki, Teddy J.; Averill, Christopher L.; Wrocklage, Kristen M.; Scott, J. Cobb; Averill, Lynnette A.; Schweinsburg, Brian; Alexander-Bloch, Aaron; Martini, Brenda; Southwick, Steven M.; Krystal, John H.; Abdallah, Chadi G. (2018). "Default mode network abnormalities in posttraumatic stress disorder: A novel network-restricted topology approach". NeuroImage. 176: 489–498. doi:10.1016/j.neuroimage.2018.05.005. ISSN 1053-8119. PMC 5976548. PMID 29730491.
- ^ Kernbach, Julius M.; Satterthwaite, Theodore D.; Bassett, Danielle S.; Smallwood, Jonathan; Margulies, Daniel; Krall, Sarah; Shaw, Philip; Varoquaux, Gaël; Thirion, Bertrand; Konrad, Kerstin; Bzdok, Danilo (17 July 2018). "Shared endo-phenotypes of default mode dysfunction in attention deficit/hyperactivity disorder and autism spectrum disorder". Translational Psychiatry. 8 (1): 133. doi:10.1038/s41398-018-0179-6. PMC 6050263. PMID 30018328.
- ^ Vigneshwaran, S; Mahanand, B. S.; Suresh, S; Sundararajan, N (2017). "Identifying differences in brain activities and an accurate detection of autism spectrum disorder using resting state functional-magnetic resonance imaging: A spatial filtering approach". Medical Image Analysis. 35: 375–389. doi:10.1016/j.media.2016.08.003. PMID 27585835. S2CID 4922560.
- ^ a b Washington, Stuart D.; Gordon, Evan M.; Brar, Jasmit; Warburton, Samantha; Sawyer, Alice T.; Wolfe, Amanda; Mease-Ference, Erin R.; Girton, Laura; Hailu, Ayichew (1 April 2014). "Dysmaturation of the default mode network in autism". Human Brain Mapping. 35 (4): 1284–1296. doi:10.1002/hbm.22252. ISSN 1097-0193. PMC 3651798. PMID 23334984.
- ^ a b Yerys, Benjamin E.; Gordon, Evan M.; Abrams, Danielle N.; Satterthwaite, Theodore D.; Weinblatt, Rachel; Jankowski, Kathryn F.; Strang, John; Kenworthy, Lauren; Gaillard, William D. (1 January 2015). "Default mode network segregation and social deficits in autism spectrum disorder: Evidence from non-medicated children". NeuroImage: Clinical. 9: 223–232. doi:10.1016/j.nicl.2015.07.018. PMC 4573091. PMID 26484047.
- ^ Meda, Shashwath A.; Ruaño, Gualberto; Windemuth, Andreas; O’Neil, Kasey; Berwise, Clifton; Dunn, Sabra M.; Boccaccio, Leah E.; Narayanan, Balaji; Kocherla, Mohan (13 May 2014). "Multivariate analysis reveals genetic associations of the resting default mode network in psychotic bipolar disorder and schizophrenia". Proceedings of the National Academy of Sciences. 111 (19): E2066–E2075. Bibcode:2014PNAS..111E2066M. doi:10.1073/pnas.1313093111. ISSN 0027-8424. PMC 4024891. PMID 24778245.
- ^ Berman, MG; Peltier, S; Nee, DE; Kross, E; Deldin, PJ; Jonides, J (October 2011). "Depression, rumination and the default network". Social Cognitive and Affective Neuroscience. 6 (5): 548–55. doi:10.1093/scan/nsq080. PMC 3190207. PMID 20855296.
- ^ Hamilton, J.Paul (2011). "Default-Mode and Task-Positive Network Activity in Major Depressive Disorder: Implications for Adaptive and Maladaptive Rumination" (PDF). Biological Psychiatry. 70 (4): 327–333. doi:10.1016/j.biopsych.2011.02.003. PMC 3144981. PMID 21459364. Retrieved 6 June 2014.
- ^ Zhu, X; Wang, X; Xiao, J; Liao, J; Zhong, M; Wang, W; Yao, S (2012). "Evidence of a dissociation pattern in resting-state default mode network connectivity in first-episode, treatment-naive major depression patients". Biological Psychiatry. 71 (7): 611–7. doi:10.1016/j.biopsych.2011.10.035. PMID 22177602. S2CID 23697809.
- ^ Kucyi, A; Moayedi, M; Weissman-Fogel, I; Goldberg, M. B.; Freeman, B. V.; Tenenbaum, H. C.; Davis, K. D. (2014). "Enhanced medial prefrontal-default mode network functional connectivity in chronic pain and its association with pain rumination". Journal of Neuroscience. 34 (11): 3969–75. doi:10.1523/JNEUROSCI.5055-13.2014. PMC 6705280. PMID 24623774.
- ^ Sambataro, Fabio; Wolf, Nadine; Giusti, Pietro; Vasic, Nenad; Wolf, Robert (October 2013). "Default mode network in depression: A pathway to impaired affective cognition?" (PDF). Clinical Neuralpyschiatry. 10: 212–216. Archived from the original (PDF) on 29 August 2017. Retrieved 28 September 2017.
- ^ Dr. Ruth Lanius, Brain Mapping conference, London, November 2010
- ^ Hoekzema, E; Carmona, S; Ramos-Quiroga, JA; Richarte Fernández, V; Bosch, R; Soliva, JC; Rovira, M; Bulbena, A; Tobeña, A; Casas, M; Vilarroya, O (April 2014). "An independent components and functional connectivity analysis of resting state fMRI data points to neural network dysregulation in adult ADHD". Human Brain Mapping. 35 (4): 1261–72. doi:10.1002/hbm.22250. PMC 6869838. PMID 23417778.
- ^ a b Mills, BD; Miranda-Dominguez, O; Mills, KL; Earl, E; Cordova, M; Painter, J; Karalunas, SL; Nigg, JT; Fair, DA (2018). "ADHD and attentional control: Impaired segregation of task positive and task negative brain networks". Network Neuroscience. 2 (2): 200–217. doi:10.1162/netn_a_00034. PMC 6130439. PMID 30215033.
- ^ Sripada, CS; Kessler, D; Angstadt, M (30 September 2014). "Lag in maturation of the brain's intrinsic functional architecture in attention-deficit/hyperactivity disorder". Proceedings of the National Academy of Sciences of the United States of America. 111 (39): 14259–64. Bibcode:2014PNAS..11114259S. doi:10.1073/pnas.1407787111. PMC 4191792. PMID 25225387.
- ^ Huang, Wenjing; Pach, Daniel; Napadow, Vitaly; Park, Kyungmo; Long, Xiangyu; Neumann, Jane; Maeda, Yumi; Nierhaus, Till; Liang, Fanrong; Witt, Claudia M.; Harrison, Ben J. (9 April 2012). "Characterizing Acupuncture Stimuli Using Brain Imaging with fMRI – A Systematic Review and Meta-Analysis of the Literature". PLOS ONE. 7 (4): e32960. Bibcode:2012PLoSO...732960H. doi:10.1371/journal.pone.0032960. PMC 3322129. PMID 22496739.
- ^ Chae, Younbyoung; Chang, Dong-Seon; Lee, Soon-Ho; Jung, Won-Mo; Lee, In-Seon; Jackson, Stephen; Kong, Jian; Lee, Hyangsook; Park, Hi-Joon; Lee, Hyejung; Wallraven, Christian (March 2013). "Inserting Needles into the Body: A Meta-Analysis of Brain Activity Associated With Acupuncture Needle Stimulation". The Journal of Pain. 14 (3): 215–222. doi:10.1016/j.jpain.2012.11.011. PMID 23395475. S2CID 36594091.
- ^ a b Akiki, Teddy J.; Averill, Christopher L.; Abdallah, Chadi G. (2017). "A Network-Based Neurobiological Model of PTSD: Evidence From Structural and Functional Neuroimaging Studies". Current Psychiatry Reports. 19 (11): 81. doi:10.1007/s11920-017-0840-4. ISSN 1523-3812. PMC 5960989. PMID 28924828.
- ^ Kowalski, Joachim; Wierzba, Małgorzata; Wypych, Marek; Marchewka, Artur; Dragan, Małgorzata (1 September 2020). "Effects of attention training technique on brain function in high- and low-cognitive-attentional syndrome individuals: Regional dynamics before, during, and after a single session of ATT". Behaviour Research and Therapy. 132: 103693. doi:10.1016/j.brat.2020.103693. ISSN 0005-7967. PMID 32688045. S2CID 220669531.
- ^ Kringelbach, Morten L.; Green, Alexander L.; Aziz, Tipu Z. (2 May 2011). "Balancing the Brain: Resting State Networks and Deep Brain Stimulation". Frontiers in Integrative Neuroscience. 5: 8. doi:10.3389/fnint.2011.00008. PMC 3088866. PMID 21577250.
- ^ a b Fox, Kieran C. R.; Nijeboer, Savannah; Dixon, Matthew L.; Floman, James L.; Ellamil, Melissa; Rumak, Samuel P.; Sedlmeier, Peter; Christoff, Kalina (2014). "Is meditation associated with altered brain structure? A systematic review and meta-analysis of morphometric neuroimaging in meditation practitioners". Neuroscience & Biobehavioral Reviews. 43: 48–73. doi:10.1016/j.neubiorev.2014.03.016. PMID 24705269. S2CID 207090878.
- ^ Raffone, Antonino; Srinivasan, Narayanan (2010). "The exploration of meditation in the neuroscience of attention and consciousness". Cognitive Processing. 11 (1): 1–7. doi:10.1007/s10339-009-0354-z. PMID 20041276.
- ^ Xu, J; Vik, A; Groote, IR; Lagopoulos, J; Holen, A; Ellingsen, Ø; Håberg, AK; Davanger, S (2014). "Nondirective meditation activates default mode network and areas associated with memory retrieval and emotional processing". Front. Hum. Neurosci. 8 (86): 86. doi:10.3389/fnhum.2014.00086. PMC 3935386. PMID 24616684.
- ^ Voss, Michelle W.; Soto, Carmen; Yoo, Seungwoo; Sodoma, Matthew; Vivar, Carmen; van Praag, Henriette (April 2019). "Exercise and Hippocampal Memory Systems". Trends in Cognitive Sciences. 23 (4): 318–333. doi:10.1016/j.tics.2019.01.006. PMC 6422697. PMID 30777641.
- ^ Shao, Mengling; Lin, Huiyan; Yin, Desheng; Li, Yongjie; Wang, Yifan; Ma, Junpeng; Yin, Jianzhong; Jin, Hua (1 October 2019). Rao, Hengyi (ed.). "Learning to play badminton altered resting-state activity and functional connectivity of the cerebellar sub-regions in adults". PLOS ONE. 14 (10): e0223234. Bibcode:2019PLoSO..1423234S. doi:10.1371/journal.pone.0223234. ISSN 1932-6203. PMC 6771995. PMID 31574108.
- ^ Muraskin, Jordan; Dodhia, Sonam; Lieberman, Gregory; Garcia, Javier O.; Verstynen, Timothy; Vettel, Jean M.; Sherwin, Jason; Sajda, Paul (December 2016). "Brain dynamics of post-task resting state are influenced by expertise: Insights from baseball players: Brain Dynamics of Post-Task Resting State". Human Brain Mapping. 37 (12): 4454–4471. doi:10.1002/hbm.23321. PMC 5113676. PMID 27448098.
- ^ Carhart-Harris, Robin L.; Erritzoe, David; Williams, Tim; Stone, James M.; Reed, Laurence J.; Colasanti, Alessandro; Tyacke, Robin J.; Leech, Robert; Malizia, Andrea L.; Murphy, Kevin; Hobden, Peter; Evans, John; Feilding, Amanda; Wise, Richard G.; Nutt, David J. (2012). "Neural correlates of the psychedelic state as determined by fMRI studies with psilocybin". PNAS. 109 (6): 2138–2143. doi:10.1073/pnas.1119598109. PMC 3277566. PMID 22308440.
- ^ Carhart-Harris, Robin L.; Muthukumaraswamy, Suresh; Roseman, Leor; Kaelen, Mendel; Droog, Wouter; Murphy, Kevin; Tagliazucchi, Enzo; Schenberg, Eduardo E.; Nest, Timothy; Orban, Csaba; Leech, Robert; Williams, Luke T.; Williams, Tim M.; Bolstridge, Mark; Sessa, Ben; McGonigle, John; Sereno, Martin I.; Nichols, David; Hellyer, Peter J.; Hobden, Peter; Evans, John; Singh, Krish D.; Wise, Richard G.; Curran, H. Valerie; Feilding, Amanda; Nutt, David J. (26 April 2016). "Neural correlates of the LSD experience revealed by multimodal neuroimaging". Proceedings of the National Academy of Sciences. 113 (17): 4853–4858. Bibcode:2016PNAS..113.4853C. doi:10.1073/pnas.1518377113. PMC 4855588. PMID 27071089.
- ^ Sripada, Rebecca K.; King, Anthony P.; Welsh, Robert C.; Garfinkel, Sarah N.; Wang, Xin; Sripada, Chandra S.; Liberzon, Israel (2012). "Neural Dysregulation in Posttraumatic Stress Disorder". Psychosomatic Medicine. 74 (9): 904–911. doi:10.1097/PSY.0b013e318273bf33. ISSN 0033-3174. PMC 3498527. PMID 23115342.
- ^ McKenna, Benjamin S.; Eyler, Lisa T. (2012). "Overlapping prefrontal systems involved in cognitive and emotional processing in euthymic bipolar disorder and following sleep deprivation: A review of functional neuroimaging studies". Clinical Psychology Review. 32 (7): 650–663. doi:10.1016/j.cpr.2012.07.003. PMC 3922056. PMID 22926687.
- ^ Basner, Mathias; Rao, Hengyi; Goel, Namni; Dinges, David F (October 2013). "Sleep deprivation and neurobehavioral dynamics". Current Opinion in Neurobiology. 23 (5): 854–863. doi:10.1016/j.conb.2013.02.008. PMC 3700596. PMID 23523374.
- ^ a b c d e Picchioni, Dante; Duyn, Jeff H.; Horovitz, Silvina G. (15 October 2013). "Sleep and the functional connectome". NeuroImage. 80: 387–396. doi:10.1016/j.neuroimage.2013.05.067. PMC 3733088. PMID 23707592.
- ^ Foster, Brett L.; Parvizi, Josef (1 March 2012). "Resting oscillations and cross-frequency coupling in the human posteromedial cortex". NeuroImage. 60 (1): 384–391. doi:10.1016/j.neuroimage.2011.12.019. ISSN 1095-9572. PMC 3596417. PMID 22227048.
- ^ Morris, Peter G.; Smith, Stephen M.; Barnes, Gareth R.; Stephenson, Mary C.; Hale, Joanne R.; Price, Darren; Luckhoo, Henry; Woolrich, Mark; Brookes, Matthew J. (4 October 2011). "Investigating the electrophysiological basis of resting state networks using magnetoencephalography". Proceedings of the National Academy of Sciences. 108 (40): 16783–16788. Bibcode:2011PNAS..10816783B. doi:10.1073/pnas.1112685108. ISSN 0027-8424. PMC 3189080. PMID 21930901.
- ^ Fair, D. A.; Cohen, A. L.; Dosenbach, N. U. F.; Church, J. A.; Miezin, F. M.; Barch, D. M.; Raichle, M. E.; Petersen, S. E.; Schlaggar, B. L. (2008). "The maturing architecture of the brain's default network". Proceedings of the National Academy of Sciences. 105 (10): 4028–32. Bibcode:2008PNAS..105.4028F. doi:10.1073/pnas.0800376105. PMC 2268790. PMID 18322013.
- ^ Morcom, Alexa M.; Fletcher, Paul C. (October 2007). "Does the brain have a baseline? Why we should be resisting a rest". NeuroImage. 37 (4): 1073–1082. doi:10.1016/j.neuroimage.2006.09.013. PMID 17052921. S2CID 3356303.
External links[edit]
Electrocorticography[edit]
| Electrocorticography | |
|---|---|
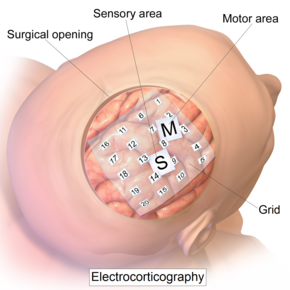 Intracranial electrode grid for electrocorticography. | |
| Synonyms | Intracranial electroencephalography |
| Purpose | record electrical activity from the cerebral cortex.(invasive) |
Electrocorticography (ECoG), a type of intracranial electroencephalography (iEEG), is a type of electrophysiological monitoring that uses electrodes placed directly on the exposed surface of the brain to record electrical activity from the cerebral cortex. In contrast, conventional electroencephalography (EEG) electrodes monitor this activity from outside the skull. ECoG may be performed either in the operating room during surgery (intraoperative ECoG) or outside of surgery (extraoperative ECoG). Because a craniotomy (a surgical incision into the skull) is required to implant the electrode grid, ECoG is an invasive procedure.
History[edit]
ECoG was pioneered in the early 1950s by Wilder Penfield and Herbert Jasper, neurosurgeons at the Montreal Neurological Institute.[1] The two developed ECoG as part of their groundbreaking Montreal procedure, a surgical protocol used to treat patients with severe epilepsy. The cortical potentials recorded by ECoG were used to identify epileptogenic zones – regions of the cortex that generate epileptic seizures. These zones would then be surgically removed from the cortex during resectioning, thus destroying the brain tissue where epileptic seizures had originated. Penfield and Jasper also used electrical stimulation during ECoG recordings in patients undergoing epilepsy surgery under local anesthesia.[2] This procedure was used to explore the functional anatomy of the brain, mapping speech areas and identifying the somatosensory and somatomotor cortex areas to be excluded from surgical removal. A doctor named Robert Galbraith Heath was also an early researcher of the brain at the Tulane University School of Medicine.[3][4]
Electrophysiological basis[edit]
ECoG signals are composed of synchronized postsynaptic potentials (local field potentials), recorded directly from the exposed surface of the cortex. The potentials occur primarily in cortical pyramidal cells, and thus must be conducted through several layers of the cerebral cortex, cerebrospinal fluid (CSF), pia mater, and arachnoid mater before reaching subdural recording electrodes placed just below the dura mater (outer cranial membrane). However, to reach the scalp electrodes of a conventional electroencephalogram (EEG), electrical signals must also be conducted through the skull, where potentials rapidly attenuate due to the low conductivity of bone. For this reason, the spatial resolution of ECoG is much higher than EEG, a critical imaging advantage for presurgical planning.[5] ECoG offers a temporal resolution of approximately 5 ms and spatial resolution as low as 1-100 µm.[6]
Using depth electrodes, the local field potential gives a measure of a neural population in a sphere with a radius of 0.5–3 mm around the tip of the electrode.[7] With a sufficiently high sampling rate (more than about 10 kHz), depth electrodes can also measure action potentials.[8] In which case the spatial resolution is down to individual neurons, and the field of view of an individual electrode is approximately 0.05–0.35 mm.[7]
Procedure[edit]
The ECoG recording is performed from electrodes placed on the exposed cortex. In order to access the cortex, a surgeon must first perform a craniotomy, removing a part of the skull to expose the brain surface. This procedure may be performed either under general anesthesia or under local anesthesia if patient interaction is required for functional cortical mapping. Electrodes are then surgically implanted on the surface of the cortex, with placement guided by the results of preoperative EEG and magnetic resonance imaging (MRI). Electrodes may either be placed outside the dura mater (epidural) or under the dura mater (subdural). ECoG electrode arrays typically consist of sixteen sterile, disposable stainless steel, carbon tip, platinum, Platinum-iridium alloy or gold ball electrodes, each mounted on a ball and socket joint for ease in positioning. These electrodes are attached to an overlying frame in a "crown" or "halo" configuration.[9] Subdural strip and grid electrodes are also widely used in various dimensions, having anywhere from 4 to 256[10] electrode contacts. The grids are transparent, flexible, and numbered at each electrode contact. Standard spacing between grid electrodes is 1 cm; individual electrodes are typically 5 mm in diameter. The electrodes sit lightly on the cortical surface, and are designed with enough flexibility to ensure that normal movements of the brain do not cause injury. A key advantage of strip and grid electrode arrays is that they may be slid underneath the dura mater into cortical regions not exposed by the craniotomy. Strip electrodes and crown arrays may be used in any combination desired. Depth electrodes may also be used to record activity from deeper structures such as the hippocampus.
DCES[edit]
Direct cortical electrical stimulation (DCES), also known as cortical stimulation mapping, is frequently performed in concurrence with ECoG recording for functional mapping of the cortex and identification of critical cortical structures.[9] When using a crown configuration, a handheld wand bipolar stimulator may be used at any location along the electrode array. However, when using a subdural strip, stimulation must be applied between pairs of adjacent electrodes due to the nonconductive material connecting the electrodes on the grid. Electrical stimulating currents applied to the cortex are relatively low, between 2 and 4 mA for somatosensory stimulation, and near 15 mA for cognitive stimulation.[9] The stimulation frequency is usually 60 Hz in North America and 50 Hz in Europe, and any charge density more than 150 μC/cm2 causes tissue damage.[11][12]
The functions most commonly mapped through DCES are primary motor, primary sensory, and language. The patient must be alert and interactive for mapping procedures, though patient involvement varies with each mapping procedure. Language mapping may involve naming, reading aloud, repetition, and oral comprehension; somatosensory mapping requires that the patient describe sensations experienced across the face and extremities as the surgeon stimulates different cortical regions.[9]
Clinical applications[edit]
Since its development in the 1950s, ECoG has been used to localize epileptogenic zones during presurgical planning, map out cortical functions, and to predict the success of epileptic surgical resectioning. ECoG offers several advantages over alternative diagnostic modalities:
- Flexible placement of recording and stimulating electrodes[2]
- Can be performed at any stage before, during, and after a surgery
- Allows for direct electrical stimulation of the brain, identifying critical regions of the cortex to be avoided during surgery
- Greater precision and sensitivity than an EEG scalp recording – spatial resolution is higher and signal-to-noise ratio is superior due to closer proximity to neural activity
Limitations of ECoG include:
- Limited sampling time – seizures (ictal events) may not be recorded during the ECoG recording period
- Limited field of view – electrode placement is limited by the area of exposed cortex and surgery time, sampling errors may occur
- Recording is subject to the influence of anesthetics, narcotic analgesics, and the surgery itself[2]
Intractable epilepsy[edit]
Epilepsy is currently ranked as the third most commonly diagnosed neurological disorder, afflicting approximately 2.5 million people in the United States alone.[13] Epileptic seizures are chronic and unrelated to any immediately treatable causes, such as toxins or infectious diseases, and may vary widely based on etiology, clinical symptoms, and site of origin within the brain. For patients with intractable epilepsy – epilepsy that is unresponsive to anticonvulsants – surgical treatment may be a viable treatment option. Partial epilepsy[14] is the common intractable epilepsy and the partial seizure is difficult to locate.Treatment for such epilepsy is limited to attachment of vagus nerve stimulator. Epilepsy surgery is the cure for partial epilepsy provided that the brain region generating seizure is carefully and accurately removed.
- Extraoperative ECoG
Before a patient can be identified as a candidate for resectioning surgery, MRI must be performed to demonstrate the presence of a structural lesion within the cortex, supported by EEG evidence of epileptogenic tissue.[2] Once a lesion has been identified, ECoG may be performed to determine the location and extent of the lesion and surrounding irritative region. The scalp EEG, while a valuable diagnostic tool, lacks the precision necessary to localize the epileptogenic region. ECoG is considered to be the gold standard for assessing neuronal activity in patients with epilepsy, and is widely used for presurgical planning to guide surgical resection of the lesion and epileptogenic zone.[15][16] The success of the surgery depends on accurate localization and removal of the epileptogenic zone. ECoG data is assessed with regard to ictal spike activity – "diffuse fast wave activity" recorded during a seizure – and interictal epileptiform activity (IEA), brief bursts of neuronal activity recorded between epileptic events. ECoG is also performed following the resectioning surgery to detect any remaining epileptiform activity, and to determine the success of the surgery. Residual spikes on the ECoG, unaltered by the resection, indicate poor seizure control, and incomplete neutralization of the epileptogenic cortical zone. Additional surgery may be necessary to completely eradicate seizure activity. Extraoperative ECoG is also used to localize functionally-important areas (also known as eloquent cortex) to be preserved during epilepsy surgery. [17] Motor, sensory, cognitive tasks during extraoperative ECoG are reported to increase the amplitude of high-frequency activity at 70–110 Hz in areas involved in execution of given tasks.[17][18][19] Task-related high-frequency activity can animate 'when' and 'where' cerebral cortex is activated and inhibited in a 4D manner with a temporal resolution of 10 milliseconds or below and a spatial resolution of 10 mm or below.[18][19]
- Intraoperative ECoG
The objective of the resectioning surgery is to remove the epileptogenic tissue without causing unacceptable neurological consequences. In addition to identifying and localizing the extent of epileptogenic zones, ECoG used in conjunction with DCES is also a valuable tool for functional cortical mapping. It is vital to precisely localize critical brain structures, identifying which regions the surgeon must spare during resectioning (the "eloquent cortex") in order to preserve sensory processing, motor coordination, and speech. Functional mapping requires that the patient be able to interact with the surgeon, and thus is performed under local rather than general anesthesia. Electrical stimulation using cortical and acute depth electrodes is used to probe distinct regions of the cortex in order to identify centers of speech, somatosensory integration, and somatomotor processing. During the resectioning surgery, intraoperative ECoG may also be performed to monitor the epileptic activity of the tissue and ensure that the entire epileptogenic zone is resectioned.
Although the use of extraoperative and intraoperative ECoG in resectioning surgery has been an accepted clinical practice for several decades, recent studies have shown that the usefulness of this technique may vary based on the type of epilepsy a patient exhibits. Kuruvilla and Flink reported that while intraoperative ECoG plays a critical role in tailored temporal lobectomies, in multiple subpial transections (MST), and in the removal of malformations of cortical development (MCDs), it has been found impractical in standard resection of medial temporal lobe epilepsy (TLE) with MRI evidence of mesial temporal sclerosis (MTS).[2] A study performed by Wennberg, Quesney, and Rasmussen demonstrated the presurgical significance of ECoG in frontal lobe epilepsy (FLE) cases.[20]
Research applications[edit]
ECoG has recently emerged as a promising recording technique for use in brain-computer interfaces (BCI).[21] BCIs are direct neural interfaces that provide control of prosthetic, electronic, or communication devices via direct use of the individual's brain signals. Brain signals may be recorded either invasively, with recording devices implanted directly into the cortex, or noninvasively, using EEG scalp electrodes. ECoG serves to provide a partially invasive compromise between the two modalities – while ECoG does not penetrate the blood–brain barrier like invasive recording devices, it features a higher spatial resolution and higher signal-to-noise ratio than EEG.[21] ECoG has gained attention recently for decoding imagined speech or music, which could lead to "literal" BCIs[22] in which users simply imagine words, sentences, or music that the BCI can directly interpret.[23][24]
In addition to clinical applications to localize functional regions to support neurosurgery, real-time functional brain mapping with ECoG has gained attention to support research into fundamental questions in neuroscience. For example, a 2017 study explored regions within face and color processing areas and found that these subregions made highly specific contributions to different aspects of vision.[25] Another study found that high-frequency activity from 70 to 200 Hz reflected processes associated with both transient and sustained decision-making.[26] Other work based on ECoG presented a new approach to interpreting brain activity, suggesting that both power and phase jointly influence instantaneous voltage potential, which directly regulates cortical excitability.[27] Like the work toward decoding imagined speech and music, these research directions involving real-time functional brain mapping also have implications for clinical practice, including both neurosurgery and BCI systems. The system that was used in most of these real-time functional mapping publications, "CortiQ". has been used for both research and clinical applications.
Recent advances[edit]
This article needs additional citations for verification. (September 2022) |
The electrocorticogram is still considered to be the "gold standard" for defining epileptogenic zones; however, this procedure is risky and highly invasive. Recent studies have explored the development of a noninvasive cortical imaging technique for presurgical planning that may provide similar information and resolution of the invasive ECoG.
In one novel approach, Lei Ding et al.[28] seek to integrate the information provided by a structural MRI and scalp EEG to provide a noninvasive alternative to ECoG. This study investigated a high-resolution subspace source localization approach, FINE (first principle vectors) to image the locations and estimate the extents of current sources from the scalp EEG. A thresholding technique was applied to the resulting tomography of subspace correlation values in order to identify epileptogenic sources. This method was tested in three pediatric patients with intractable epilepsy, with encouraging clinical results. Each patient was evaluated using structural MRI, long-term video EEG monitoring with scalp electrodes, and subsequently with subdural electrodes. The ECoG data were then recorded from implanted subdural electrode grids placed directly on the surface of the cortex. MRI and computed tomography images were also obtained for each subject.
The epileptogenic zones identified from preoperative EEG data were validated by observations from postoperative ECoG data in all three patients. These preliminary results suggest that it is possible to direct surgical planning and locate epileptogenic zones noninvasively using the described imaging and integrating methods. EEG findings were further validated by the surgical outcomes of all three patients. After surgical resectioning, two patients are seizure-free and the third has experienced a significant reduction in seizures. Due to its clinical success, FINE offers a promising alternative to preoperative ECoG, providing information about both the location and extent of epileptogenic sources through a noninvasive imaging procedure.
See also[edit]
References[edit]
- ^ Palmini, A (2006). "The concept of the epileptogenic zone: A modern look at Penfield and Jasper's views on the role of interictal spikes". Epileptic Disorders. 8 (Suppl 2): S10–5. doi:10.1684/j.1950-6945.2006.tb00205.x. hdl:10923/21709. PMID 17012068.
- ^ a b c d e Kuruvilla, A; Flink, R (2003). "Intraoperative electrocorticography in epilepsy surgery: Useful or not?". Seizure. 12 (8): 577–84. doi:10.1016/S1059-1311(03)00095-5. PMID 14630497. S2CID 15643130.
- ^ Baumeister AA (2000). "The Tulane Electrical Brain Stimulation Program a historical case study in medical ethics". J Hist Neurosci. 9 (3): 262–78. doi:10.1076/jhin.9.3.262.1787. PMID 11232368. S2CID 38336466.
- ^ Marwan Hariz; Patric Blomstedt; Ludvic Zrinzo (2016). "Deep Brain Stimulation between 1947 and 1987: The Untold Story". Neurosurg Focus. 29 (2). e1 – via Medscape.
- ^ Hashiguchi, K; Morioka, T; Yoshida, F; Miyagi, Y; et al. (2007). "Correlation between scalp-recorded electroencephalographic and electrocorticographic activities during ictal period". Seizure. 16 (3): 238–247. doi:10.1016/j.seizure.2006.12.010. PMID 17236792. S2CID 1728557.
- ^ Fallegger, Florian; Schiavone, Giuseppe; Pirondini, Elvira; Wagner, Fabien B.; Vachicouras, Nicolas; Serex, Ludovic; Zegarek, Gregory; May, Adrien; Constanthin, Paul; Palma, Marie; Khoshnevis, Mehrdad; Van Roost, Dirk; Yvert, Blaise; Courtine, Grégoire; Schaller, Karl (March 2021). "MRI-Compatible and Conformal Electrocorticography Grids for Translational Research". Advanced Science. 8 (9). doi:10.1002/advs.202003761. ISSN 2198-3844. PMC 8097365. PMID 33977054.
- ^ a b Logothetis, NK (2003). "The underpinning of the BOLD functional magnetic resonance imaging signal". The Journal of Neuroscience. 23 (10): 3963–71. doi:10.1523/JNEUROSCI.23-10-03963.2003. PMC 6741096. PMID 12764080.
- ^ Ulbert, I; Halgren, E; Heit, G; Karmos, G (2001). "Multiple microelectrode-recording system for human intracortical applications". Journal of Neuroscience Methods. 106 (1): 69–79. doi:10.1016/S0165-0270(01)00330-2. PMID 11248342. S2CID 12203755.
- ^ a b c d Schuh, L; Drury, I (1996). "Intraoperative electrocorticography and direct cortical electrical stimulation". Seminars in Anesthesia. 16: 46–55. doi:10.1016/s0277-0326(97)80007-4.
- ^ Mesgarani, N; Chang, EF (2012). "Selective cortical representation of attended speaker in multi-talker speech perception". Nature. 485 (7397): 233–6. Bibcode:2012Natur.485..233M. doi:10.1038/nature11020. PMC 3870007. PMID 22522927.
- ^ Boyer A, Duffau H, Vincent M, Ramdani S, Mandonnet E, Guiraud D, Bonnetblanc F (2018). "Electrophysiological Activity Evoked by Direct Electrical Stimulation of the Human Brain: Interest of the P0 Component" (PDF). 2018 40th Annual International Conference of the IEEE Engineering in Medicine and Biology Society (EMBC). Vol. 2018. pp. 2210–2213. doi:10.1109/EMBC.2018.8512733. ISBN 978-1-5386-3646-6. PMID 30440844. S2CID 53097668.
- ^ Ritaccio, Anthony L.; Brunner, Peter; Schalk, Gerwin (March 2018). "Electrical Stimulation Mapping of the Brain: Basic Principles and Emerging Alternatives". Journal of Clinical Neurophysiology. 35 (2): 86–97. doi:10.1097/WNP.0000000000000440. ISSN 0736-0258. PMC 5836484. PMID 29499015."frequency" on page 6, "damage" on page 3 of pdf
- ^ Kohrman, M (2007). "What is epilepsy? Clinical perspectives in the diagnosis and treatment". Journal of Clinical Neurophysiology. 24 (2): 87–95. doi:10.1097/WNP.0b013e3180415b51. PMID 17414964. S2CID 35146214.
- ^ Wetjen, Nicholas M.; Marsh, W. Richard; Meyer, Fredric B.; Cascino, Gregory D.; So, Elson; Britton, Jeffrey W.; Stead, S. Matthew; Worrell, Gregory A. (June 2009). "Intracranial electroencephalography seizure onset patterns and surgical outcomes in nonlesional extratemporal epilepsy: Clinical article". Journal of Neurosurgery. 110 (6): 1147–1152. doi:10.3171/2008.8.JNS17643. ISSN 0022-3085. PMC 2841508. PMID 19072306.
- ^ Sugano, H; Shimizu, H; Sunaga, S (2007). "Efficacy of intraoperative electrocorticography for assessing seizure outcomes in intractable epilepsy patients with temporal-lobe-mass lesions". Seizure. 16 (2): 120–127. doi:10.1016/j.seizure.2006.10.010. PMID 17158074.
- ^ Miller, KJ; denNijs, M; Shenoy, P; Miller, JW; et al. (2007). "Real-time functional brain mapping using electrocorticography". NeuroImage. 37 (2): 504–507. doi:10.1016/j.neuroimage.2007.05.029. PMID 17604183. S2CID 3362496.
- ^ a b Crone, NE; Miglioretti, DL; Gordon, B; Lesser, RP (1998). "Functional mapping of human sensorimotor cortex with electrocorticographic spectral analysis. II. Event-related synchronization in the gamma band". Brain. 121 (12): 2301–15. doi:10.1093/brain/121.12.2301. PMID 9874481.
- ^ a b Nakai, Y; Jeong, JW; Brown, EC; Rothermel, R; Kojima, K; Kambara, T; Shah, A; Mittal, S; Sood, S; Asano, E (2017). "Three- and four-dimensional mapping of speech and language in patients with epilepsy". Brain. 140 (5): 1351–1370. doi:10.1093/brain/awx051. PMC 5405238. PMID 28334963.
- ^ a b Nakai, Ya; Nagashima, A; Hayakawa, A; Osuki, T; Jeong, JW; Sugiura, A; Brown, EC; Asano, E (2018). "Four-dimensional map of the human early visual system". Clin Neurophysiol. 129 (1): 188–197. doi:10.1016/j.clinph.2017.10.019. PMC 5743586. PMID 29190524.
- ^ Wennberg, R; Quesney, F; Olivier, A; Rasmussen, T (1998). "Electrocorticography and outcome in frontal lobe epilepsy". Electroencephalography and Clinical Neurophysiology. 106 (4): 357–68. doi:10.1016/S0013-4694(97)00148-X. PMID 9741764.
- ^ a b Shenoy, P; Miller, KJ; Ojemann, JG; Rao, RPN (2007). "Generalized features for electrocorticographic BCIs" (PDF). IEEE Transactions on Biomedical Engineering. 55 (1): 273–80. CiteSeerX 10.1.1.208.7298. doi:10.1109/TBME.2007.903528. PMID 18232371. S2CID 3034381. Archived (PDF) from the original on 2014-12-14.
- ^ Allison, Brendan Z. (2009). "Chapter 2: Toward Ubiquitous BCIs.". Brain-Computer Interfaces. Springer. pp. 357–387. ISBN 978-3-642-02091-9.
- ^ Swift, James; Coon, William; Guger, Christoph; Brunner, Peter; Bunch, M; Lynch, T; Frawley, T; Ritaccio, Anthony; Schalk, Gerwin (2018). "Passive functional mapping of receptive language areas using electrocorticographic signals". Clinical Neurophysiology. 6 (12): 2517–2524. doi:10.1016/j.clinph.2018.09.007. PMC 6414063. PMID 30342252.
- ^ Martin, Stephanie; Iturrate, Iñaki; Millán, José del R.; Knight, Robert; Pasley, Brian N. (2018). "Decoding Inner Speech Using Electrocorticography: Progress and Challenges Toward a Speech Prosthesis". Frontiers in Neuroscience. 12: 422. doi:10.3389/fnins.2018.00422. PMC 6021529. PMID 29977189.
- ^ Schalk, Gerwin; Kapeller, Christoph; Guger, Christoph; Ogawa, H; Hiroshima, S; Lafer-Sousa, R; Saygin, Zenyip M.; Kamada, Kyousuke; Kanwisher, Nancy (2017). "Facephenes and rainbows: Causal evidence for functional and anatomical specificity of face and color processing in the human brain". Proc Natl Acad Sci U S A. 114 (46): 12285–12290. Bibcode:2017PNAS..11412285S. doi:10.1073/pnas.1713447114. PMC 5699078. PMID 29087337.
- ^ Saez, I; Lin, J; Stolk, A; Chang, E; Parvizi, J; Schalk, Gerwin; Knight, Robert T.; Hsu, M. (2018). "Encoding of Multiple Reward-Related Computations in Transient and Sustained High-Frequency Activity in Human OFC". Current Biology. 28 (18): 2889–2899.e3. doi:10.1016/j.cub.2018.07.045. PMC 6590063. PMID 30220499.
- ^ Schalk, Gerwin; Marple, J.; Knight, Robert T.; Coon, William G. (2017). "Instantaneous voltage as an alternative to power- and phase-based interpretation of oscillatory brain activity". NeuroImage. 157: 545–554. doi:10.1016/j.neuroimage.2017.06.014. PMC 5600843. PMID 28624646.
- ^ Ding, L; Wilke, C; Xu, B; Xu, X; et al. (2007). "EEG source imaging: Correlating source locations and extents with electrocorticography and surgical resections in epilepsy patients". Journal of Clinical Neurophysiology. 24 (2): 130–136. doi:10.1097/WNP.0b013e318038fd52. PMC 2758789. PMID 17414968.
Brain–computer interface[edit]
This article may require copy editing for grammar, style, cohesion, tone, or spelling. (April 2024) |
| Neuropsychology |
|---|
 |
A brain–computer interface (BCI), sometimes called a brain–machine interface (BMI), is a direct communication pathway between the brain's electrical activity and an external device, most commonly a computer or robotic limb. BCIs are often directed at researching, mapping, assisting, augmenting, or repairing human cognitive or sensory-motor functions.[1] They are often conceptualized as a human–machine interface that skips the intermediary component of the physical movement of body parts, although they also raise the possibility of the erasure of the discreteness of brain and machine. Implementations of BCIs range from non-invasive (EEG, MEG, MRI) and partially invasive (ECoG and endovascular) to invasive (microelectrode array), based on how close electrodes get to brain tissue.[2]
Research on BCIs began in the 1970s by Jacques Vidal at the University of California, Los Angeles (UCLA) under a grant from the National Science Foundation, followed by a contract from DARPA.[3][4] Vidal's 1973 paper marks the first appearance of the expression brain–computer interface in scientific literature.
Due to the cortical plasticity of the brain, signals from implanted prostheses can, after adaptation, be handled by the brain like natural sensor or effector channels.[5] Following years of animal experimentation, the first neuroprosthetic devices implanted in humans appeared in the mid-1990s.
Recently, studies in human-computer interaction via the application of machine learning to statistical temporal features extracted from the frontal lobe (EEG brainwave) data has had high levels of success in classifying mental states (relaxed, neutral, concentrating),[6] mental emotional states (negative, neutral, positive),[7] and thalamocortical dysrhythmia.[8]
History[edit]
The history of brain–computer interfaces (BCIs) starts with Hans Berger's discovery of the electrical activity of the human brain and the development of electroencephalography (EEG). In 1924 Berger was the first to record human brain activity by means of EEG. Berger was able to identify oscillatory activity, such as Berger's wave or the alpha wave (8–13 Hz), by analyzing EEG traces.
Berger's first recording device was very rudimentary. He inserted silver wires under the scalps of his patients. These were later replaced by silver foils attached to the patient's head by rubber bandages. Berger connected these sensors to a Lippmann capillary electrometer, with disappointing results. However, more sophisticated measuring devices, such as the Siemens double-coil recording galvanometer, which displayed electric voltages as small as one ten thousandth of a volt, led to success.
Berger analyzed the interrelation of alternations in his EEG wave diagrams with brain diseases. EEGs permitted completely new possibilities for the research of human brain activities.
Although the term had not yet been coined, one of the earliest examples of a working brain-machine interface was the piece Music for Solo Performer (1965) by the American composer Alvin Lucier. The piece makes use of EEG and analog signal processing hardware (filters, amplifiers, and a mixing board) to stimulate acoustic percussion instruments. To perform the piece one must produce alpha waves and thereby "play" the various percussion instruments via loudspeakers which are placed near or directly on the instruments themselves.[9]
UCLA Professor Jacques Vidal coined the term "BCI" and produced the first peer-reviewed publications on this topic.[3][4] Vidal is widely recognized as the inventor of BCIs in the BCI community, as reflected in numerous peer-reviewed articles reviewing and discussing the field (e.g.,[10][11][12]). A review pointed out that Vidal's 1973 paper stated the "BCI challenge"[13] of controlling external objects using EEG signals, and especially use of Contingent Negative Variation (CNV) potential as a challenge for BCI control. The 1977 experiment Vidal described was the first application of BCI after his 1973 BCI challenge. It was a noninvasive EEG (actually Visual Evoked Potentials (VEP)) control of a cursor-like graphical object on a computer screen. The demonstration was movement in a maze.[14]
After his early contributions, Vidal was not active in BCI research, nor BCI events such as conferences, for many years. In 2011, however, he gave a lecture in Graz, Austria, supported by the Future BNCI project, presenting the first BCI, which earned a standing ovation. Vidal was joined by his wife, Laryce Vidal, who previously worked with him at UCLA on his first BCI project.
In 1988, a report was given on noninvasive EEG control of a physical object, a robot. The experiment described was EEG control of multiple start-stop-restart of the robot movement, along an arbitrary trajectory defined by a line drawn on a floor. The line-following behavior was the default robot behavior, utilizing autonomous intelligence and autonomous source of energy.[15][16] This 1988 report written by Stevo Bozinovski, Mihail Sestakov, and Liljana Bozinovska was the first one about a robot control using EEG.[17][18]
In 1990, a report was given on a closed loop, bidirectional adaptive BCI controlling computer buzzer by an anticipatory brain potential, the Contingent Negative Variation (CNV) potential.[19][20] The experiment described how an expectation state of the brain, manifested by CNV, controls in a feedback loop the S2 buzzer in the S1-S2-CNV paradigm. The obtained cognitive wave representing the expectation learning in the brain is named Electroexpectogram (EXG). The CNV brain potential was part of the BCI challenge presented by Vidal in his 1973 paper.
Studies in 2010s suggested the potential ability of neural stimulation to restore functional connectively and associated behaviors through modulation of molecular mechanisms of synaptic efficacy.[21][22] This opened the door for the concept that BCI technologies may be able to restore function in addition to enabling functionality.
Since 2013, DARPA has funded BCI technology through the BRAIN initiative, which has supported work out of the University of Pittsburgh Medical Center,[23] Paradromics,[24] Brown,[25] and Synchron,[26] among others.
Versus neuroprosthetics[edit]
Neuroprosthetics is an area of neuroscience concerned with neural prostheses, that is, using artificial devices to replace the function of impaired nervous systems and brain-related problems, or of sensory organs or organs itself (bladder, diaphragm, etc.). As of December 2010, cochlear implants had been implanted as neuroprosthetic device in approximately 736,900 people worldwide.[27] There are also several neuroprosthetic devices that aim to restore vision, including retinal implants. The first neuroprosthetic device, however, was the pacemaker.
The terms are sometimes used interchangeably. Neuroprosthetics and BCIs seek to achieve the same aims, such as restoring sight, hearing, movement, ability to communicate, and even cognitive function.[1] Both use similar experimental methods and surgical techniques.
Animal BCI research[edit]
Several laboratories have managed to record signals from monkey and rat cerebral cortices to operate BCIs to produce movement. Monkeys have navigated computer cursors on screen and commanded robotic arms to perform simple tasks simply by thinking about the task and seeing the visual feedback, but without any motor output.[28] In May 2008 photographs that showed a monkey at the University of Pittsburgh Medical Center operating a robotic arm by thinking were published in a number of well-known science journals and magazines.[29] Sheep too have been used to evaluate BCI technology including Synchron's Stentrode.
In 2020, Elon Musk's Neuralink was successfully implanted in a pig,[30] announced in a widely viewed webcast. In 2021, Elon Musk announced that he had successfully enabled a monkey to play video games using Neuralink's device.[31]
Early work[edit]
In 1969 the operant conditioning studies of Fetz and colleagues, at the Regional Primate Research Center and Department of Physiology and Biophysics, University of Washington School of Medicine in Seattle, showed for the first time that monkeys could learn to control the deflection of a biofeedback meter arm with neural activity.[32] Similar work in the 1970s established that monkeys could quickly learn to voluntarily control the firing rates of individual and multiple neurons in the primary motor cortex if they were rewarded for generating appropriate patterns of neural activity.[33]
Studies that developed algorithms to reconstruct movements from motor cortex neurons, which control movement, date back to the 1970s. In the 1980s, Apostolos Georgopoulos at Johns Hopkins University found a mathematical relationship between the electrical responses of single motor cortex neurons in rhesus macaque monkeys and the direction in which they moved their arms (based on a cosine function). He also found that dispersed groups of neurons, in different areas of the monkey's brains, collectively controlled motor commands, but was able to record the firings of neurons in only one area at a time, because of the technical limitations imposed by his equipment.[34]
There has been rapid development in BCIs since the mid-1990s.[35] Several groups have been able to capture complex brain motor cortex signals by recording from neural ensembles (groups of neurons) and using these to control external devices.
Prominent research successes[edit]
Kennedy and Yang Dan[edit]
Phillip Kennedy (who later founded Neural Signals in 1987) and colleagues built the first intracortical brain–computer interface by implanting neurotrophic-cone electrodes into monkeys.[citation needed]
In 1999, researchers led by Yang Dan at the University of California, Berkeley decoded neuronal firings to reproduce images seen by cats. The team used an array of electrodes embedded in the thalamus (which integrates all of the brain's sensory input) of sharp-eyed cats. Researchers targeted 177 brain cells in the thalamus lateral geniculate nucleus area, which decodes signals from the retina. The cats were shown eight short movies, and their neuron firings were recorded. Using mathematical filters, the researchers decoded the signals to generate movies of what the cats saw and were able to reconstruct recognizable scenes and moving objects.[36] Similar results in humans have since been achieved by researchers in Japan (see below).
Nicolelis[edit]
Miguel Nicolelis, a professor at Duke University, in Durham, North Carolina, has been a prominent proponent of using multiple electrodes spread over a greater area of the brain to obtain neuronal signals to drive a BCI.
After conducting initial studies in rats during the 1990s, Nicolelis and his colleagues developed BCIs that decoded brain activity in owl monkeys and used the devices to reproduce monkey movements in robotic arms. Monkeys have advanced reaching and grasping abilities and good hand manipulation skills, making them ideal test subjects for this kind of work.
By 2000, the group succeeded in building a BCI that reproduced owl monkey movements while the monkey operated a joystick or reached for food.[37] The BCI operated in real time and could also control a separate robot remotely over Internet Protocol. But the monkeys could not see the arm moving and did not receive any feedback, a so-called open-loop BCI.

Later experiments by Nicolelis using rhesus monkeys succeeded in closing the feedback loop and reproduced monkey reaching and grasping movements in a robot arm. With their deeply cleft and furrowed brains, rhesus monkeys are considered to be better models for human neurophysiology than owl monkeys. The monkeys were trained to reach and grasp objects on a computer screen by manipulating a joystick while corresponding movements by a robot arm were hidden.[38][39] The monkeys were later shown the robot directly and learned to control it by viewing its movements. The BCI used velocity predictions to control reaching movements and simultaneously predicted handgripping force. In 2011 O'Doherty and colleagues showed a BCI with sensory feedback with rhesus monkeys. The monkey was brain controlling the position of an avatar arm while receiving sensory feedback through direct intracortical stimulation (ICMS) in the arm representation area of the sensory cortex.[40]
Donoghue, Schwartz and Andersen[edit]

Other laboratories which have developed BCIs and algorithms that decode neuron signals include the Carney Institute for Brain Science at Brown University and the labs of Andrew Schwartz at the University of Pittsburgh and Richard Andersen at Caltech. These researchers have been able to produce working BCIs, even using recorded signals from far fewer neurons than did Nicolelis (15–30 neurons versus 50–200 neurons).
John Donoghue's lab at the Carney Institute reported training rhesus monkeys to use a BCI to track visual targets on a computer screen (closed-loop BCI) with or without assistance of a joystick.[41] Schwartz's group created a BCI for three-dimensional tracking in virtual reality and also reproduced BCI control in a robotic arm.[42] The same group also created headlines when they demonstrated that a monkey could feed itself pieces of fruit and marshmallows using a robotic arm controlled by the animal's own brain signals.[43][44][45]
Andersen's group used recordings of premovement activity from the posterior parietal cortex in their BCI, including signals created when experimental animals anticipated receiving a reward.[46]
Other research[edit]
In addition to predicting kinematic and kinetic parameters of limb movements, BCIs that predict electromyographic or electrical activity of the muscles of primates are being developed.[47] Such BCIs could be used to restore mobility in paralyzed limbs by electrically stimulating muscles.
Miguel Nicolelis and colleagues demonstrated that the activity of large neural ensembles can predict arm position. This work made possible creation of BCIs that read arm movement intentions and translate them into movements of artificial actuators. Carmena and colleagues[38] programmed the neural coding in a BCI that allowed a monkey to control reaching and grasping movements by a robotic arm. Lebedev and colleagues[39] argued that brain networks reorganize to create a new representation of the robotic appendage in addition to the representation of the animal's own limbs.
In 2019, researchers from UCSF published a study where they demonstrated a BCI that had the potential to help patients with speech impairment caused by neurological disorders. Their BCI used high-density electrocorticography to tap neural activity from a patient's brain and used deep learning methods to synthesize speech.[48][49] In 2021, researchers from the same group published a study showing the potential of a BCI to decode words and sentences in an anarthric patient who had been unable to speak for over 15 years.[50][51]
The biggest impediment to BCI technology at present is the lack of a sensor modality that provides safe, accurate and robust access to brain signals. It is conceivable or even likely, however, that such a sensor will be developed within the next twenty years. The use of such a sensor should greatly expand the range of communication functions that can be provided using a BCI.
Development and implementation of a BCI system is complex and time-consuming. In response to this problem, Gerwin Schalk has been developing a general-purpose system for BCI research, called BCI2000. BCI2000 has been in development since 2000 in a project led by the Brain–Computer Interface R&D Program at the Wadsworth Center of the New York State Department of Health in Albany, New York, United States.[52]
A new 'wireless' approach uses light-gated ion channels such as Channelrhodopsin to control the activity of genetically defined subsets of neurons in vivo. In the context of a simple learning task, illumination of transfected cells in the somatosensory cortex influenced the decision-making process of freely moving mice.[53]
The use of BMIs has also led to a deeper understanding of neural networks and the central nervous system. Research has shown that despite the inclination of neuroscientists to believe that neurons have the most effect when working together, single neurons can be conditioned through the use of BMIs to fire at a pattern that allows primates to control motor outputs. The use of BMIs has led to development of the single neuron insufficiency principle which states that even with a well tuned firing rate single neurons can only carry a narrow amount of information and therefore the highest level of accuracy is achieved by recording firings of the collective ensemble. Other principles discovered with the use of BMIs include the neuronal multitasking principle, the neuronal mass principle, the neural degeneracy principle, and the plasticity principle.[54]
BCIs are also proposed to be applied by users without disabilities. A user-centered categorization of BCI approaches by Thorsten O. Zander and Christian Kothe introduces the term passive BCI.[55] Next to active and reactive BCI that are used for directed control, passive BCIs allow for assessing and interpreting changes in the user state during Human-Computer Interaction (HCI). In a secondary, implicit control loop the computer system adapts to its user improving its usability in general.
Beyond BCI systems that decode neural activity to drive external effectors, BCI systems may be used to encode signals from the periphery. These sensory BCI devices enable real-time, behaviorally-relevant decisions based upon closed-loop neural stimulation.[56]
The BCI Award[edit]
The Annual BCI Research Award is awarded in recognition of outstanding and innovative research in the field of Brain-Computer Interfaces. Each year, a renowned research laboratory is asked to judge the submitted projects. The jury consists of world-leading BCI experts recruited by the awarding laboratory. The jury selects twelve nominees, then chooses a first, second, and third-place winner, who receive awards of $3,000, $2,000, and $1,000, respectively.
Human BCI research[edit]
Invasive BCIs[edit]
Invasive BCI requires surgery to implant electrodes under the scalp for communicating brain signals. The main advantage is to provide more accurate reading; however, its downside includes side effects from the surgery including scar tissue, which can make brain signals weaker. In addition, according to the research of Abdulkader et al., (2015),[57] the body may not accept the implanted electrodes and this can cause a medical condition.
Vision[edit]
Invasive BCI research has targeted repairing damaged sight and providing new functionality for people with paralysis. Invasive BCIs are implanted directly into the grey matter of the brain during neurosurgery. Because they lie in the grey matter, invasive devices produce the highest quality signals of BCI devices but are prone to scar-tissue build-up, causing the signal to become weaker, or even non-existent, as the body reacts to a foreign object in the brain.[58]
In vision science, direct brain implants have been used to treat non-congenital (acquired) blindness. One of the first scientists to produce a working brain interface to restore sight was private researcher William Dobelle.
Dobelle's first prototype was implanted into "Jerry", a man blinded in adulthood, in 1978. A single-array BCI containing 68 electrodes was implanted onto Jerry's visual cortex and succeeded in producing phosphenes, the sensation of seeing light. The system included cameras mounted on glasses to send signals to the implant. Initially, the implant allowed Jerry to see shades of grey in a limited field of vision at a low frame-rate. This also required him to be hooked up to a mainframe computer, but shrinking electronics and faster computers made his artificial eye more portable and now enable him to perform simple tasks unassisted.[59]

In 2002, Jens Naumann, also blinded in adulthood, became the first in a series of 16 paying patients to receive Dobelle's second generation implant, marking one of the earliest commercial uses of BCIs. The second generation device used a more sophisticated implant enabling better mapping of phosphenes into coherent vision. Phosphenes are spread out across the visual field in what researchers call "the starry-night effect". Immediately after his implant, Jens was able to use his imperfectly restored vision to drive an automobile slowly around the parking area of the research institute.[60] Unfortunately, Dobelle died in 2004[61] before his processes and developments were documented. Subsequently, when Mr. Naumann and the other patients in the program began having problems with their vision, there was no relief and they eventually lost their "sight" again. Naumann wrote about his experience with Dobelle's work in Search for Paradise: A Patient's Account of the Artificial Vision Experiment[62] and has returned to his farm in Southeast Ontario, Canada, to resume his normal activities.[63]
Movement[edit]
BCIs focusing on motor neuroprosthetics aim to either restore movement in individuals with paralysis or provide devices to assist them, such as interfaces with computers or robot arms.
Researchers at Emory University in Atlanta, led by Philip Kennedy and Roy Bakay, were first to install a brain implant in a human that produced signals of high enough quality to simulate movement. Their patient, Johnny Ray (1944–2002), developed 'locked-in syndrome' after having a brain-stem stroke in 1997. Ray's implant was installed in 1998 and he lived long enough to start working with the implant, eventually learning to control a computer cursor; he died in 2002 of a brain aneurysm.[64]
Tetraplegic Matt Nagle became the first person to control an artificial hand using a BCI in 2005 as part of the first nine-month human trial of Cyberkinetics's BrainGate chip-implant. Implanted in Nagle's right precentral gyrus (area of the motor cortex for arm movement), the 96-electrode BrainGate implant allowed Nagle to control a robotic arm by thinking about moving his hand as well as a computer cursor, lights and TV.[65] One year later, professor Jonathan Wolpaw received the prize of the Altran Foundation for Innovation to develop a Brain Computer Interface with electrodes located on the surface of the skull, instead of directly in the brain.[66]
More recently, research teams led by the BrainGate group at Brown University and a group led by University of Pittsburgh Medical Center, both in collaborations with the United States Department of Veterans Affairs, have demonstrated further success in direct control of robotic prosthetic limbs with many degrees of freedom using direct connections to arrays of neurons in the motor cortex of patients with tetraplegia.[67][68]
Communication[edit]
In May 2021, a Stanford University team reported a successful proof-of-concept test that enabled a quadraplegic participant to input English sentences at about 86 characters per minute and 18 words per minute. The participant imagined moving his hand to write letters, and the system performed handwriting recognition on electrical signals detected in the motor cortex, utilizing hidden Markov models and recurrent neural networks for decoding.[69][70]
A report published in July 2021 reported a paralyzed patient was able to communicate 15 words per minute using a brain implant that analyzed motor neurons that previously controlled the vocal tract.[71][50]
In a recent review article, researchers raised an open question of whether human information transfer rates can surpass that of language with BCIs. Given that recent language research has demonstrated that human information transfer rates are relatively constant across many languages, there may exist a limit at the level of information processing in the brain. On the contrary, this "upper limit" of information transfer rate may be intrinsic to language itself, as a modality for information transfer.[72]
In 2023 two studies used BCIs with recurrent neural network to decode speech at a record rate of 62 words per minute and 78 words per minute.[73][74][75]
Technical challenges[edit]
There exist a number of technical challenges to recording brain activity with invasive BCIs. Advances in CMOS technology are pushing and enabling integrated, invasive BCI designs with smaller size, lower power requirements, and higher signal acquisition capabilities.[76] Invasive BCIs involve electrodes that penetrate brain tissue in an attempt to record action potential signals (also known as spikes) from individual, or small groups of, neurons near the electrode. The interface between a recording electrode and the electrolytic solution surrounding neurons has been modelled using the Hodgkin-Huxley model.[77][78]
Electronic limitations to invasive BCIs have been an active area of research in recent decades. While intracellular recordings of neurons reveal action potential voltages on the scale of hundreds of millivolts, chronic invasive BCIs rely on recording extracellular voltages which typically are three orders of magnitude smaller, existing at hundreds of microvolts.[79] Further adding to the challenge of detecting signals on the scale of microvolts is the fact that the electrode-tissue interface has a high capacitance at small voltages. Due to the nature of these small signals, for BCI systems that incorporate functionality onto an integrated circuit, each electrode requires its own amplifier and ADC, which convert analog extracellular voltages into digital signals.[79] Because a typical neuron action potential lasts for one millisecond, BCIs measuring spikes must have sampling rates ranging from 300 Hz to 5 kHz. Yet another concern is that invasive BCIs must be low-power, so as to dissipate less heat to surrounding tissue; at the most basic level more power is traditionally needed to optimize signal-to-noise ratio.[78] Optimal battery design is an active area of research in BCIs.[80]
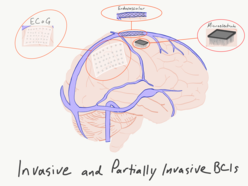
Challenges existing in the area of material science are central to the design of invasive BCIs. Variations in signal quality over time have been commonly observed with implantable microelectrodes.[81][82] Optimal material and mechanical characteristics for long term signal stability in invasive BCIs has been an active area of research.[83] It has been proposed that the formation of glial scarring, secondary to damage at the electrode-tissue interface, is likely responsible for electrode failure and reduced recording performance.[84] Research has suggested that blood-brain barrier leakage, either at the time of insertion or over time, may be responsible for the inflammatory and glial reaction to chronic microelectrodes implanted in the brain.[84][85] As a result, flexible[86][87][88] and tissue-like designs[89][90] have been researched and developed to minimize foreign-body reaction by means of matching the Young's modulus of the electrode closer to that of brain tissue.[89]
Partially invasive BCIs[edit]
Partially invasive BCI devices are implanted inside the skull but rest outside the brain rather than within the grey matter. They produce better resolution signals than non-invasive BCIs where the bone tissue of the cranium deflects and deforms signals and have a lower risk of forming scar-tissue in the brain than fully invasive BCIs. There has been preclinical demonstration of intracortical BCIs from the stroke perilesional cortex.[91]
Endovascular[edit]
A systematic review published in 2020 detailed multiple studies, both clinical and non-clinical, dating back decades investigating the feasibility of endovascular BCIs.[92]
In recent years, the biggest advance in partially invasive BCIs has emerged in the area of interventional neurology.[2] In 2010, researchers affiliated with University of Melbourne had begun developing a BCI that could be inserted via the vascular system. The Australian neurologist Thomas Oxley (Mount Sinai Hospital) conceived the idea for this BCI, called Stentrode, which has received funding from DARPA. Preclinical studies evaluated the technology in sheep.
The Stentrode, a monolithic stent electrode array, is designed to be delivered via an intravenous catheter under image-guidance to the superior sagittal sinus, in the region which lies adjacent to motor cortex.[93] This proximity to motor cortex underlies the Stentrode's ability to measure neural activity. The procedure is most similar to how venous sinus stents are placed for the treatment of idiopathic intracranial hypertension.[94] The Stentrode communicates neural activity to a battery-less telemetry unit implanted in the chest, which communicates wirelessly with an external telemetry unit capable of power and data transfer. While an endovascular BCI benefits from avoiding craniotomy for insertion, risks such as clotting and venous thrombosis are possible.
First-in-human trials with the Stentrode are underway.[93] In November 2020, two participants with amyotrophic lateral sclerosis were able to wirelessly control an operating system to text, email, shop, and bank using direct thought through the Stentrode brain-computer interface,[95] marking the first time a brain-computer interface was implanted via the patient's blood vessels, eliminating the need for open brain surgery. In January 2023, researchers reported no serious adverse events during the first year for all four patients who could use it to operate computers.[96][97]
ECoG[edit]
Electrocorticography (ECoG) measures the electrical activity of the brain taken from beneath the skull in a similar way to non-invasive electroencephalography, but the electrodes are embedded in a thin plastic pad that is placed above the cortex, beneath the dura mater.[98] ECoG technologies were first trialled in humans in 2004 by Eric Leuthardt and Daniel Moran from Washington University in St. Louis. In a later trial, the researchers enabled a teenage boy to play Space Invaders using his ECoG implant.[99] This research indicates that control is rapid, requires minimal training, and may be an ideal tradeoff with regards to signal fidelity and level of invasiveness.[note 1]
Signals can be either subdural or epidural, but are not taken from within the brain parenchyma itself. It has not been studied extensively until recently due to the limited access of subjects. Currently, the only manner to acquire the signal for study is through the use of patients requiring invasive monitoring for localization and resection of an epileptogenic focus.
ECoG is a very promising intermediate BCI modality because it has higher spatial resolution, better signal-to-noise ratio, wider frequency range, and less training requirements than scalp-recorded EEG, and at the same time has lower technical difficulty, lower clinical risk, and may have superior long-term stability than intracortical single-neuron recording.[101] This feature profile and recent evidence of the high level of control with minimal training requirements shows potential for real world application for people with motor disabilities.[102][103] Light reactive imaging BCI devices are still in the realm of theory.
Recent work published by Edward Chang and Joseph Makin from UCSF revealed that ECoG signals could be used to decode speech from epilepsy patients implanted with high-density ECoG arrays over the peri-Sylvian cortices.[104][105] Their study achieved word error rates of 3% (a marked improvement from prior publications) utilizing an encoder-decoder neural network, which translated ECoG data into one of fifty sentences composed of 250 unique words.
Non-invasive BCIs[edit]
There have also been experiments in humans using non-invasive neuroimaging technologies as interfaces. The substantial majority of published BCI work involves noninvasive EEG-based BCIs. Noninvasive EEG-based technologies and interfaces have been used for a much broader variety of applications. Although EEG-based interfaces are easy to wear and do not require surgery, they have relatively poor spatial resolution and cannot effectively use higher-frequency signals because the skull dampens signals, dispersing and blurring the electromagnetic waves created by the neurons. EEG-based interfaces also require some time and effort prior to each usage session, whereas non-EEG-based ones, as well as invasive ones require no prior-usage training. Overall, the best BCI for each user depends on numerous factors.
Functional near-infrared spectroscopy[edit]
In 2014 and 2017, a BCI using functional near-infrared spectroscopy for "locked-in" patients with amyotrophic lateral sclerosis (ALS) was able to restore some basic ability of the patients to communicate with other people.[106][107]
Electroencephalography (EEG)-based brain-computer interfaces[edit]

After the BCI challenge was stated by Vidal in 1973, the initial reports on non-invasive approach included control of a cursor in 2D using VEP (Vidal 1977), control of a buzzer using CNV (Bozinovska et al. 1988, 1990), control of a physical object, a robot, using a brain rhythm (alpha) (Bozinovski et al. 1988), control of a text written on a screen using P300 (Farwell and Donchin, 1988).[13]
In the early days of BCI research, another substantial barrier to using electroencephalography (EEG) as a brain–computer interface was the extensive training required before users can work the technology. For example, in experiments beginning in the mid-1990s, Niels Birbaumer at the University of Tübingen in Germany trained severely paralysed people to self-regulate the slow cortical potentials in their EEG to such an extent that these signals could be used as a binary signal to control a computer cursor.[108] (Birbaumer had earlier trained epileptics to prevent impending fits by controlling this low voltage wave.) The experiment saw ten patients trained to move a computer cursor by controlling their brainwaves. The process was slow, requiring more than an hour for patients to write 100 characters with the cursor, while training often took many months. However, the slow cortical potential approach to BCIs has not been used in several years, since other approaches require little or no training, are faster and more accurate, and work for a greater proportion of users.
Another research parameter is the type of oscillatory activity that is measured. Gert Pfurtscheller founded the BCI Lab 1991 and fed his research results on motor imagery in the first online BCI based on oscillatory features and classifiers. Together with Birbaumer and Jonathan Wolpaw at New York State University they focused on developing technology that would allow users to choose the brain signals they found easiest to operate a BCI, including mu and beta rhythms.
A further parameter is the method of feedback used and this is shown in studies of P300 signals. Patterns of P300 waves are generated involuntarily (stimulus-feedback) when people see something they recognize and may allow BCIs to decode categories of thoughts without training patients first. By contrast, the biofeedback methods described above require learning to control brainwaves so the resulting brain activity can be detected.
In 2005 it was reported research on EEG emulation of digital control circuits for BCI, with example of a CNV flip-flop.[109] In 2009 it was reported noninvasive EEG control of a robotic arm using a CNV flip-flop.[110] In 2011 it was reported control of two robotic arms solving Tower of Hanoi task with three disks using a CNV flip-flop.[111] In 2015 it was described EEG-emulation of a Schmitt trigger, flip-flop, demultiplexer, and modem.[112]
While an EEG based brain-computer interface has been pursued extensively by a number of research labs, recent advancements made by Bin He and his team at the University of Minnesota suggest the potential of an EEG based brain-computer interface to accomplish tasks close to invasive brain-computer interface. Using advanced functional neuroimaging including BOLD functional MRI and EEG source imaging, Bin He and co-workers identified the co-variation and co-localization of electrophysiological and hemodynamic signals induced by motor imagination.[113] Refined by a neuroimaging approach and by a training protocol, Bin He and co-workers demonstrated the ability of a non-invasive EEG based brain-computer interface to control the flight of a virtual helicopter in 3-dimensional space, based upon motor imagination.[114] In June 2013 it was announced that Bin He had developed the technique to enable a remote-control helicopter to be guided through an obstacle course.[115]
In addition to a brain-computer interface based on brain waves, as recorded from scalp EEG electrodes, Bin He and co-workers explored a virtual EEG signal-based brain-computer interface by first solving the EEG inverse problem and then used the resulting virtual EEG for brain-computer interface tasks. Well-controlled studies suggested the merits of such a source analysis based brain-computer interface.[116]
A 2014 study found that severely motor-impaired patients could communicate faster and more reliably with non-invasive EEG BCI, than with any muscle-based communication channel.[117]
A 2016 study found that the Emotiv EPOC device may be more suitable for control tasks using the attention/meditation level or eye blinking than the Neurosky MindWave device.[118]
A 2019 study found that the application of evolutionary algorithms could improve EEG mental state classification with a non-invasive Muse device, enabling high quality classification of data acquired by a cheap consumer-grade EEG sensing device.[119]
In a 2021 systematic review of randomized controlled trials using BCI for upper-limb rehabilitation after stroke, EEG-based BCI was found to have significant efficacy in improving upper-limb motor function compared to control therapies. More specifically, BCI studies that utilized band power features, motor imagery, and functional electrical stimulation in their design were found to be more efficacious than alternatives.[120] Another 2021 systematic review focused on robotic-assisted EEG-based BCI for hand rehabilitation after stroke. Improvement in motor assessment scores was observed in three of eleven studies included in the systematic review.[121]
Dry active electrode arrays[edit]
In the early 1990s Babak Taheri, at University of California, Davis demonstrated the first single and also multichannel dry active electrode arrays using micro-machining. The single channel dry EEG electrode construction and results were published in 1994.[122] The arrayed electrode was also demonstrated to perform well compared to silver/silver chloride electrodes. The device consisted of four sites of sensors with integrated electronics to reduce noise by impedance matching. The advantages of such electrodes are: (1) no electrolyte used, (2) no skin preparation, (3) significantly reduced sensor size, and (4) compatibility with EEG monitoring systems. The active electrode array is an integrated system made of an array of capacitive sensors with local integrated circuitry housed in a package with batteries to power the circuitry. This level of integration was required to achieve the functional performance obtained by the electrode.
The electrode was tested on an electrical test bench and on human subjects in four modalities of EEG activity, namely: (1) spontaneous EEG, (2) sensory event-related potentials, (3) brain stem potentials, and (4) cognitive event-related potentials. The performance of the dry electrode compared favorably with that of the standard wet electrodes in terms of skin preparation, no gel requirements (dry), and higher signal-to-noise ratio.[123]
In 1999 researchers at Case Western Reserve University, in Cleveland, Ohio, led by Hunter Peckham, used 64-electrode EEG skullcap to return limited hand movements to quadriplegic Jim Jatich. As Jatich concentrated on simple but opposite concepts like up and down, his beta-rhythm EEG output was analysed using software to identify patterns in the noise. A basic pattern was identified and used to control a switch: Above average activity was set to on, below average off. As well as enabling Jatich to control a computer cursor the signals were also used to drive the nerve controllers embedded in his hands, restoring some movement.[124]
SSVEP mobile EEG BCIs[edit]
In 2009, the NCTU Brain-Computer-Interface-headband was reported. The researchers who developed this BCI-headband also engineered silicon-based microelectro-mechanical system (MEMS) dry electrodes designed for application in non-hairy sites of the body. These electrodes were secured to the DAQ board in the headband with snap-on electrode holders. The signal processing module measured alpha activity and the Bluetooth enabled phone assessed the patients' alertness and capacity for cognitive performance. When the subject became drowsy, the phone sent arousing feedback to the operator to rouse them. This research was supported by the National Science Council, Taiwan, R.O.C., NSC, National Chiao-Tung University, Taiwan's Ministry of Education, and the U.S. Army Research Laboratory.[125]
In 2011, researchers reported a cellular based BCI with the capability of taking EEG data and converting it into a command to cause the phone to ring. This research was supported in part by Abraxis Bioscience LLP, the U.S. Army Research Laboratory, and the Army Research Office. The developed technology was a wearable system composed of a four channel bio-signal acquisition/amplification module, a wireless transmission module, and a Bluetooth enabled cell phone. The electrodes were placed so that they pick up steady state visual evoked potentials (SSVEPs).[126] SSVEPs are electrical responses to flickering visual stimuli with repetition rates over 6 Hz[126] that are best found in the parietal and occipital scalp regions of the visual cortex.[127][128][129] It was reported that with this BCI setup, all study participants were able to initiate the phone call with minimal practice in natural environments.[130]
The scientists claim that their studies using a single channel fast Fourier transform (FFT) and multiple channel system canonical correlation analysis (CCA) algorithm support the capacity of mobile BCIs.[126][131] The CCA algorithm has been applied in other experiments investigating BCIs with claimed high performance in accuracy as well as speed.[132] While the cellular based BCI technology was developed to initiate a phone call from SSVEPs, the researchers said that it can be translated for other applications, such as picking up sensorimotor mu/beta rhythms to function as a motor-imagery based BCI.[126]
In 2013, comparative tests were performed on android cell phone, tablet, and computer based BCIs, analyzing the power spectrum density of resultant EEG SSVEPs. The stated goals of this study, which involved scientists supported in part by the U.S. Army Research Laboratory, were to "increase the practicability, portability, and ubiquity of an SSVEP-based BCI, for daily use". Citation It was reported that the stimulation frequency on all mediums was accurate, although the cell phone's signal demonstrated some instability. The amplitudes of the SSVEPs for the laptop and tablet were also reported to be larger than those of the cell phone. These two qualitative characterizations were suggested as indicators of the feasibility of using a mobile stimulus BCI.[131]
Limitations[edit]
In 2011, researchers stated that continued work should address ease of use, performance robustness, reducing hardware and software costs.[126]
One of the difficulties with EEG readings is the large susceptibility to motion artifacts.[133] In most of the previously described research projects, the participants were asked to sit still, reducing head and eye movements as much as possible, and measurements were taken in a laboratory setting. However, since the emphasized application of these initiatives had been in creating a mobile device for daily use,[131] the technology had to be tested in motion.
In 2013, researchers tested mobile EEG-based BCI technology, measuring SSVEPs from participants as they walked on a treadmill at varying speeds. This research was supported by the Office of Naval Research, Army Research Office, and the U.S. Army Research Laboratory. Stated results were that as speed increased the SSVEP detectability using CCA decreased. As independent component analysis (ICA) had been shown to be efficient in separating EEG signals from noise,[134] the scientists applied ICA to CCA extracted EEG data. They stated that the CCA data with and without ICA processing were similar. Thus, they concluded that CCA independently demonstrated a robustness to motion artifacts that indicates it may be a beneficial algorithm to apply to BCIs used in real world conditions.[128] One of the major problems in EEG-based BCI applications is the low spatial resolution. Several solutions have been suggested to address this issue since 2019, which include: EEG source connectivity based on graph theory, EEG pattern recognition based on Topomap, EEG-fMRI fusion, and so on.
Prosthesis and environment control[edit]
Non-invasive BCIs have also been applied to enable brain-control of prosthetic upper and lower extremity devices in people with paralysis. For example, Gert Pfurtscheller of Graz University of Technology and colleagues demonstrated a BCI-controlled functional electrical stimulation system to restore upper extremity movements in a person with tetraplegia due to spinal cord injury.[135] Between 2012 and 2013, researchers at the University of California, Irvine demonstrated for the first time that it is possible to use BCI technology to restore brain-controlled walking after spinal cord injury. In their spinal cord injury research study, a person with paraplegia was able to operate a BCI-robotic gait orthosis to regain basic brain-controlled ambulation.[136][137] In 2009 Alex Blainey, an independent researcher based in the UK, successfully used the Emotiv EPOC to control a 5 axis robot arm.[138] He then went on to make several demonstration mind controlled wheelchairs and home automation that could be operated by people with limited or no motor control such as those with paraplegia and cerebral palsy.
Research into military use of BCIs funded by DARPA has been ongoing since the 1970s.[3][4] The current focus of research is user-to-user communication through analysis of neural signals.[139]
MEG and MRI[edit]
Magnetoencephalography (MEG) and functional magnetic resonance imaging (fMRI) have both been used successfully as non-invasive BCIs.[140] In a widely reported experiment, fMRI allowed two users being scanned to play Pong in real-time by altering their haemodynamic response or brain blood flow through biofeedback techniques.[141]
fMRI measurements of haemodynamic responses in real time have also been used to control robot arms with a seven-second delay between thought and movement.[142]
In 2008 research developed in the Advanced Telecommunications Research (ATR) Computational Neuroscience Laboratories in Kyoto, Japan, allowed the scientists to reconstruct images directly from the brain and display them on a computer in black and white at a resolution of 10x10 pixels. The article announcing these achievements was the cover story of the journal Neuron of 10 December 2008.[143]
In 2011 researchers from UC Berkeley published[144] a study reporting second-by-second reconstruction of videos watched by the study's subjects, from fMRI data. This was achieved by creating a statistical model relating visual patterns in videos shown to the subjects, to the brain activity caused by watching the videos. This model was then used to look up the 100 one-second video segments, in a database of 18 million seconds of random YouTube videos, whose visual patterns most closely matched the brain activity recorded when subjects watched a new video. These 100 one-second video extracts were then combined into a mashed-up image that resembled the video being watched.[145][146][147]
BCI control strategies in neurogaming[edit]
Motor imagery[edit]
Motor imagery involves the imagination of the movement of various body parts resulting in sensorimotor cortex activation, which modulates sensorimotor oscillations in the EEG. This can be detected by the BCI to infer a user's intent. Motor imagery typically requires a number of sessions of training before acceptable control of the BCI is acquired. These training sessions may take a number of hours over several days before users can consistently employ the technique with acceptable levels of precision. Regardless of the duration of the training session, users are unable to master the control scheme. This results in very slow pace of the gameplay.[148] Advanced machine learning methods were recently developed to compute a subject-specific model for detecting the performance of motor imagery. The top performing algorithm from BCI Competition IV[149] dataset 2 for motor imagery is the Filter Bank Common Spatial Pattern, developed by Ang et al. from A*STAR, Singapore.[150]
Bio/neurofeedback for passive BCI designs[edit]
Biofeedback is used to monitor a subject's mental relaxation. In some cases, biofeedback does not monitor electroencephalography (EEG), but instead bodily parameters such as electromyography (EMG), galvanic skin resistance (GSR), and heart rate variability (HRV). Many biofeedback systems are used to treat certain disorders such as attention deficit hyperactivity disorder (ADHD), sleep problems in children, teeth grinding, and chronic pain. EEG biofeedback systems typically monitor four different bands (theta: 4–7 Hz, alpha:8–12 Hz, SMR: 12–15 Hz, beta: 15–18 Hz) and challenge the subject to control them. Passive BCI[55] involves using BCI to enrich human–machine interaction with implicit information on the actual user's state, for example, simulations to detect when users intend to push brakes during an emergency car stopping procedure. Game developers using passive BCIs need to acknowledge that through repetition of game levels the user's cognitive state will change or adapt. Within the first play of a level, the user will react to things differently from during the second play: for example, the user will be less surprised at an event in the game if they are expecting it.[148]
Visual evoked potential (VEP)[edit]
A VEP is an electrical potential recorded after a subject is presented with a type of visual stimuli. There are several types of VEPs.
Steady-state visually evoked potentials (SSVEPs) use potentials generated by exciting the retina, using visual stimuli modulated at certain frequencies. SSVEP's stimuli are often formed from alternating checkerboard patterns and at times simply use flashing images. The frequency of the phase reversal of the stimulus used can be clearly distinguished in the spectrum of an EEG; this makes detection of SSVEP stimuli relatively easy. SSVEP has proved to be successful within many BCI systems. This is due to several factors, the signal elicited is measurable in as large a population as the transient VEP and blink movement and electrocardiographic artefacts do not affect the frequencies monitored. In addition, the SSVEP signal is exceptionally robust; the topographic organization of the primary visual cortex is such that a broader area obtains afferents from the central or fovial region of the visual field. SSVEP does have several problems however. As SSVEPs use flashing stimuli to infer a user's intent, the user must gaze at one of the flashing or iterating symbols in order to interact with the system. It is, therefore, likely that the symbols could become irritating and uncomfortable to use during longer play sessions, which can often last more than an hour which may not be an ideal gameplay.
Another type of VEP used with applications is the P300 potential. The P300 event-related potential is a positive peak in the EEG that occurs at roughly 300 ms after the appearance of a target stimulus (a stimulus for which the user is waiting or seeking) or oddball stimuli. The P300 amplitude decreases as the target stimuli and the ignored stimuli grow more similar.The P300 is thought to be related to a higher level attention process or an orienting response using P300 as a control scheme has the advantage of the participant only having to attend limited training sessions. The first application to use the P300 model was the P300 matrix. Within this system, a subject would choose a letter from a grid of 6 by 6 letters and numbers. The rows and columns of the grid flashed sequentially and every time the selected "choice letter" was illuminated the user's P300 was (potentially) elicited. However, the communication process, at approximately 17 characters per minute, was quite slow. The P300 is a BCI that offers a discrete selection rather than a continuous control mechanism. The advantage of P300 use within games is that the player does not have to teach himself/herself how to use a completely new control system and so only has to undertake short training instances, to learn the gameplay mechanics and basic use of the BCI paradigm.[148]
Non-brain-based human–computer interface (physiological computing)[edit]
Human-computer interaction can benefit from other recording modalities, such as EOG and eye-tracking. However, these modalities do not record brain activity and therefore do not fall within the exact scope of BCIs, but rather can be grouped under the wider field of physiological computing.[151]
Electro-oculography (EOG)[edit]
In 1989, a report was given on control of a mobile robot by eye movement using electrooculography (EOG) signals. A mobile robot was driven from a start to a goal point using five EOG commands, interpreted as forward, backward, left, right, and stop.[152]
Pupil-size oscillation[edit]
A 2016 article[153] described an entirely new communication device and non-EEG-based human-computer interface, which requires no visual fixation, or ability to move the eyes at all. The interface is based on covert interest; directing one's attention to a chosen letter on a virtual keyboard, without the need to move one's eyes to look directly at the letter. Each letter has its own (background) circle which micro-oscillates in brightness differently from all of the other letters. The letter selection is based on best fit between unintentional pupil-size oscillation and the background circle's brightness oscillation pattern. Accuracy is additionally improved by the user's mental rehearsing of the words 'bright' and 'dark' in synchrony with the brightness transitions of the letter's circle.
Synthetic telepathy[edit]
In a $6.3 million US Army initiative to invent devices for telepathic communication, Gerwin Schalk, underwritten in a $2.2 million grant, found the use of ECoG signals can discriminate the vowels and consonants embedded in spoken and imagined words, shedding light on the distinct mechanisms associated with production of vowels and consonants, and could provide the basis for brain-based communication using imagined speech.[103][154]
In 2002 Kevin Warwick had an array of 100 electrodes fired into his nervous system in order to link his nervous system into the Internet to investigate enhancement possibilities. With this in place Warwick successfully carried out a series of experiments. With electrodes also implanted into his wife's nervous system, they conducted the first direct electronic communication experiment between the nervous systems of two humans.[155][156][157][158]
Another group of researchers was able to achieve conscious brain-to-brain communication between two people separated by a distance using non-invasive technology that was in contact with the scalp of the participants. The words were encoded by binary streams using the sequences of 0's and 1's by the imaginary motor input of the person "emitting" the information. As the result of this experiment, pseudo-random bits of the information carried encoded words "hola" ("hi" in Spanish) and "ciao" ("goodbye" in Italian) and were transmitted mind-to-mind between humans separated by a distance, with blocked motor and sensory systems, which has low to no probability of this happening by chance.[159]
In the 1960s a researcher was successful after some training in using EEG to create Morse code using their brain alpha waves. Research funded by the US army is being conducted with the goal of allowing users to compose a message in their head, then transfer that message with just the power of thought to a particular individual.[160] On 27 February 2013 the group with Miguel Nicolelis at Duke University and IINN-ELS successfully connected the brains of two rats with electronic interfaces that allowed them to directly share information, in the first-ever direct brain-to-brain interface.[161][162][163]
Cell-culture BCIs[edit]
Researchers have built devices to interface with neural cells and entire neural networks in cultures outside animals. As well as furthering research on animal implantable devices, experiments on cultured neural tissue have focused on building problem-solving networks, constructing basic computers and manipulating robotic devices. Research into techniques for stimulating and recording from individual neurons grown on semiconductor chips is sometimes referred to as neuroelectronics or neurochips.[164]
Development of the first working neurochip was claimed by a Caltech team led by Jerome Pine and Michael Maher in 1997.[165] The Caltech chip had room for 16 neurons.
In 2003 a team led by Theodore Berger, at the University of Southern California, started work on a neurochip designed to function as an artificial or prosthetic hippocampus. The neurochip was designed to function in rat brains and was intended as a prototype for the eventual development of higher-brain prosthesis. The hippocampus was chosen because it is thought to be the most ordered and structured part of the brain and is the most studied area. Its function is to encode experiences for storage as long-term memories elsewhere in the brain.[166]
In 2004 Thomas DeMarse at the University of Florida used a culture of 25,000 neurons taken from a rat's brain to fly a F-22 fighter jet aircraft simulator.[167] After collection, the cortical neurons were cultured in a petri dish and rapidly began to reconnect themselves to form a living neural network. The cells were arranged over a grid of 60 electrodes and used to control the pitch and yaw functions of the simulator. The study's focus was on understanding how the human brain performs and learns computational tasks at a cellular level.
Collaborative BCIs[edit]
The idea of combining/integrating brain signals from multiple individuals was introduced at Humanity+ @Caltech, in December 2010, by a Caltech researcher at JPL, Adrian Stoica, who referred to the concept as multi-brain aggregation.[168][169][170] A provisional patent application was filed on January 19, 2011, with the non-provisional patent following one year later.[171] In May 2011, Yijun Wang and Tzyy-Ping Jung published, "A Collaborative Brain-Computer Interface for Improving Human Performance", and in January 2012 Miguel Eckstein published, "Neural decoding of collective wisdom with multi-brain computing".[172][173] Stoica's first paper on the topic appeared in 2012, after the publication of his patent application.[174] Given the timing of the publications between the patent and papers, Stoica, Wang & Jung, and Eckstein independently pioneered the concept, and are all considered as founders of the field. Later, Stoica would collaborate with University of Essex researchers, Riccardo Poli and Caterina Cinel.[175][176] The work was continued by Poli and Cinel, and their students: Ana Matran-Fernandez, Davide Valeriani, and Saugat Bhattacharyya.[177][178][179]
Ethical considerations[edit]
As technology continually blurs the line between science fiction and reality, the advent of brain-computer interfaces (BCIs) poses a profound ethical quandary. These neural interfaces, heralded as marvels of innovation, facilitate direct communication between the human brain and external devices. However, the ethical landscape surrounding BCIs is intricate and multifaceted, encompassing concerns of privacy invasion, autonomy, consent, and the potential societal implications of merging human cognition with machine interfaces. Delving into the ethical considerations of BCIs illuminates the intricate balance between technological advancement and safeguarding fundamental human rights and values. Many of the concerns raised can be divided into two groups, user centric issues and legal and social issues.
Ethical concerns in the user centric sphere tend to revolve around the safety of the user and the effects that this technology will have on them over a period of time. These can include but are not limited to: long-term effects to the user remain largely unknown, obtaining informed consent from people who have difficulty communicating, the consequences of BCI technology for the quality of life of patients and their families, health-related side-effects (e.g. neurofeedback of sensorimotor rhythm training is reported to affect sleep quality), therapeutic applications and their potential misuse, safety risks, non-convertibility of some of the changes made to the brain, lack of access to maintenance, repair and spare parts in case of company bankruptcy,[180] etc.
The legal and social aspect of BCIs is a metaphorical minefield for any entity attempting to make BCIs mainstream. Some of these concerns would be issues of accountability and responsibility: claims that the influence of BCIs overrides free will and control over sensory-motor actions, claims that cognitive intention was inaccurately translated due to a BCI malfunction, personality changes involved caused by deep-brain stimulation, concerns regarding the state of becoming a "cyborg" - having parts of the body that are living and parts that are mechanical, questions about personality: what does it mean to be a human, blurring of the division between human and machine and inability to distinguish between human vs. machine-controlled actions,[181] use of the technology in advanced interrogation techniques by governmental authorities, “brain hacking” or the unauthorized access of someones BCI,[182] selective enhancement and social stratification, mind reading and privacy, tracking and "tagging system", mind control, movement control, and emotion control.[183] In addition many researchers have theorized that BCIs would only worsen social inequalities seen today.
In their current form, most BCIs are far removed from the ethical issues considered above. They are actually similar to corrective therapies in function. Clausen stated in 2009 that "BCIs pose ethical challenges, but these are conceptually similar to those that bioethicists have addressed for other realms of therapy[184]". Moreover, he suggests that bioethics is well-prepared to deal with the issues that arise with BCI technologies. Haselager and colleagues[185] pointed out that expectations of BCI efficacy and value play a great role in ethical analysis and the way BCI scientists should approach media. Furthermore, standard protocols can be implemented to ensure ethically sound informed-consent procedures with locked-in patients.
The case of BCIs today has parallels in medicine, as will its evolution. Similar to how pharmaceutical science began as a balance for impairments and is now used to increase focus and reduce need for sleep, BCIs will likely transform gradually from therapies to enhancements.[186] Efforts are made inside the BCI community to create consensus on ethical guidelines for BCI research, development and dissemination.[187] As innovation continues, ensuring equitable access to BCIs will be crucial, failing which generational inequalities can arise which can adversely affect the right to human flourishing.
Low-cost BCI-based interfaces[edit]
Recently a number of companies have scaled back medical grade EEG technology to create inexpensive BCIs for research as well as entertainment purposes. For example, toys such as the NeuroSky and Mattel MindFlex have seen some commercial success.
- In 2006 Sony patented a neural interface system allowing radio waves to affect signals in the neural cortex.[188]
- In 2007 NeuroSky released the first affordable consumer based EEG along with the game NeuroBoy. This was also the first large scale EEG device to use dry sensor technology.[189]
- In 2008 OCZ Technology developed a device for use in video games relying primarily on electromyography.[190]
- In 2008 Final Fantasy developer Square Enix announced that it was partnering with NeuroSky to create a game, Judecca.[191][192]
- In 2009 Mattel partnered with NeuroSky to release the Mindflex, a game that used an EEG to steer a ball through an obstacle course. It is by far the best selling consumer based EEG to date.[191][193]
- In 2009 Uncle Milton Industries partnered with NeuroSky to release the Star Wars Force Trainer, a game designed to create the illusion of possessing the Force.[191][194]
- In 2009 Emotiv released the EPOC, a 14 channel EEG device that can read 4 mental states, 13 conscious states, facial expressions, and head movements. The EPOC is the first commercial BCI to use dry sensor technology, which can be dampened with a saline solution for a better connection.[195]
- In November 2011 Time magazine selected "necomimi" produced by Neurowear as one of the best inventions of the year. The company announced that it expected to launch a consumer version of the garment, consisting of catlike ears controlled by a brain-wave reader produced by NeuroSky, in spring 2012.[196]
- In February 2014 They Shall Walk (a nonprofit organization fixed on constructing exoskeletons, dubbed LIFESUITs, for paraplegics and quadriplegics) began a partnership with James W. Shakarji on the development of a wireless BCI.[197]
- In 2016, a group of hobbyists developed an open-source BCI board that sends neural signals to the audio jack of a smartphone, dropping the cost of entry-level BCI to £20.[198] Basic diagnostic software is available for Android devices, as well as a text entry app for Unity.[199]
- In 2020, NextMind released a dev kit including an EEG headset with dry electrodes at $399.[200][201] The device can be played with some demo applications or developers can create their own use cases using the provided Software Development Kit.
Future directions[edit]

A consortium consisting of 12 European partners has completed a roadmap to support the European Commission in their funding decisions for the new framework program Horizon 2020. The project, which was funded by the European Commission, started in November 2013 and published a roadmap in April 2015.[202] A 2015 publication led by Clemens Brunner describes some of the analyses and achievements of this project, as well as the emerging Brain-Computer Interface Society.[203] For example, this article reviewed work within this project that further defined BCIs and applications, explored recent trends, discussed ethical issues, and evaluated different directions for new BCIs.
Other recent publications too have explored future BCI directions for new groups of disabled users (e.g.,[10][204])
Disorders of consciousness (DOC)[edit]
Some people have a disorder of consciousness (DOC). This state is defined to include people in a coma and those in a vegetative state (VS) or minimally conscious state (MCS). New BCI research seeks to help people with DOC in different ways. A key initial goal is to identify patients who can perform basic cognitive tasks, which would of course lead to a change in their diagnosis. That is, some people who are diagnosed with DOC may in fact be able to process information and make important life decisions (such as whether to seek therapy, where to live, and their views on end-of-life decisions regarding them). Some who are diagnosed with DOC die as a result of end-of-life decisions, which may be made by family members who sincerely feel this is in the patient's best interests. Given the new prospect of allowing these patients to provide their views on this decision, there would seem to be a strong ethical pressure to develop this research direction to guarantee that DOC patients are given an opportunity to decide whether they want to live.[205][206]
These and other articles describe new challenges and solutions to use BCI technology to help persons with DOC. One major challenge is that these patients cannot use BCIs based on vision. Hence, new tools rely on auditory and/or vibrotactile stimuli. Patients may wear headphones and/or vibrotactile stimulators placed on the wrists, neck, leg, and/or other locations. Another challenge is that patients may fade in and out of consciousness and can only communicate at certain times. This may indeed be a cause of mistaken diagnosis. Some patients may only be able to respond to physicians' requests for a few hours per day (which might not be predictable ahead of time) and thus may have been unresponsive during diagnosis. Therefore, new methods rely on tools that are easy to use in field settings, even without expert help, so family members and other people without any medical or technical background can still use them. This reduces the cost, time, need for expertise, and other burdens with DOC assessment. Automated tools can ask simple questions that patients can easily answer, such as "Is your father named George?" or "Were you born in the USA?" Automated instructions inform patients that they may convey yes or no by (for example) focusing their attention on stimuli on the right vs. left wrist. This focused attention produces reliable changes in EEG patterns that can help determine whether the patient is able to communicate. The results could be presented to physicians and therapists, which could lead to a revised diagnosis and therapy. In addition, these patients could then be provided with BCI-based communication tools that could help them convey basic needs, adjust bed position and HVAC (heating, ventilation, and air conditioning), and otherwise empower them to make major life decisions and communicate.[207][208][209]
Motor recovery[edit]
People may lose some of their ability to move due to many causes, such as stroke or injury. Research in recent years has demonstrated the utility of EEG-based BCI systems in aiding motor recovery and neurorehabilitation in patients who have had a stroke.[210][211][212][213] Several groups have explored systems and methods for motor recovery that include BCIs.[214][215][216][217] In this approach, a BCI measures motor activity while the patient imagines or attempts movements as directed by a therapist. The BCI may provide two benefits: (1) if the BCI indicates that a patient is not imagining a movement correctly (non-compliance), then the BCI could inform the patient and therapist; and (2) rewarding feedback such as functional stimulation or the movement of a virtual avatar also depends on the patient's correct movement imagery.
So far, BCIs for motor recovery have relied on the EEG to measure the patient's motor imagery. However, studies have also used fMRI to study different changes in the brain as persons undergo BCI-based stroke rehab training.[218][219][220] Imaging studies combined with EEG-based BCI systems hold promise for investigating neuroplasticity during motor recovery post-stroke.[220] Future systems might include the fMRI and other measures for real-time control, such as functional near-infrared, probably in tandem with EEGs. Non-invasive brain stimulation has also been explored in combination with BCIs for motor recovery.[221] In 2016, scientists out of the University of Melbourne published preclinical proof-of-concept data related to a potential brain-computer interface technology platform being developed for patients with paralysis to facilitate control of external devices such as robotic limbs, computers and exoskeletons by translating brain activity.[222][223] Clinical trials are currently underway.[224]
Functional brain mapping[edit]
Each year, about 400,000 people undergo brain mapping during neurosurgery. This procedure is often required for people with tumors or epilepsy that do not respond to medication.[225] During this procedure, electrodes are placed on the brain to precisely identify the locations of structures and functional areas. Patients may be awake during neurosurgery and asked to perform certain tasks, such as moving fingers or repeating words. This is necessary so that surgeons can remove only the desired tissue while sparing other regions, such as critical movement or language regions. Removing too much brain tissue can cause permanent damage, while removing too little tissue can leave the underlying condition untreated and require additional neurosurgery.[citation needed] Thus, there is a strong need to improve both methods and systems to map the brain as effectively as possible.
In several recent publications, BCI research experts and medical doctors have collaborated to explore new ways to use BCI technology to improve neurosurgical mapping. This work focuses largely on high gamma activity, which is difficult to detect with non-invasive means. Results have led to improved methods for identifying key areas for movement, language, and other functions. A recent article addressed advances in functional brain mapping and summarizes a workshop.[226]
Flexible devices[edit]
Flexible electronics are polymers or other flexible materials (e.g. silk,[227] pentacene, PDMS, Parylene, polyimide[228]) that are printed with circuitry; the flexible nature of the organic background materials allowing the electronics created to bend, and the fabrication techniques used to create these devices resembles those used to create integrated circuits and microelectromechanical systems (MEMS).[citation needed] Flexible electronics were first developed in the 1960s and 1970s, but research interest increased in the mid-2000s.[229]
Flexible neural interfaces have been extensively tested in recent years in an effort to minimize brain tissue trauma related to mechanical mismatch between electrode and tissue.[230] Minimizing tissue trauma could, in theory, extend the lifespan of BCIs relying on flexible electrode-tissue interfaces.
Neural dust[edit]
Neural dust is a term used to refer to millimeter-sized devices operated as wirelessly powered nerve sensors that were proposed in a 2011 paper from the University of California, Berkeley Wireless Research Center, which described both the challenges and outstanding benefits of creating a long lasting wireless BCI.[231][232] In one proposed model of the neural dust sensor, the transistor model allowed for a method of separating between local field potentials and action potential "spikes", which would allow for a greatly diversified wealth of data acquirable from the recordings.[231]
See also[edit]
- Informatics
- Intendix (2009)
- AlterEgo, a system that reads unspoken verbalizations and responds with bone-conduction headphones
- Augmented learning
- Biological machine
- Cortical implants
- Deep brain stimulation
- Human senses
- Experience machine
- Kernel (neurotechnology company)
- Lie detection
- Microwave auditory effect
- Neural engineering
- Neuralink
- Neurorobotics
- Neurostimulation
- Nootropic
- Project Cyborg
- Simulated reality
- Telepresence
- Thought identification
- Wetware computer (Uses similar technology for IO)
- Whole brain emulation
- Wirehead (science fiction)
Notes[edit]
References[edit]
- ^ a b Krucoff MO, Rahimpour S, Slutzky MW, Edgerton VR, Turner DA (2016). "Enhancing Nervous System Recovery through Neurobiologics, Neural Interface Training, and Neurorehabilitation". Frontiers in Neuroscience. 10: 584. doi:10.3389/fnins.2016.00584. PMC 5186786. PMID 28082858.
- ^ a b Michael L Martini, BA, Eric Karl Oermann, MD, Nicholas L Opie, PhD, Fedor Panov, MD, Thomas Oxley, MD, PhD, Kurt Yaeger, MD, Sensor Modalities for Brain-Computer Interface Technology: A Comprehensive Literature Review, Neurosurgery, Volume 86, Issue 2, February 2020, Pages E108–E117, [1]
- ^ a b c Vidal JJ (1973). "Toward direct brain-computer communication". Annual Review of Biophysics and Bioengineering. 2 (1): 157–180. doi:10.1146/annurev.bb.02.060173.001105. PMID 4583653.
- ^ a b c Vidal J (1977). "Real-Time Detection of Brain Events in EEG". Proceedings of the IEEE. 65 (5): 633–641. doi:10.1109/PROC.1977.10542. S2CID 7928242.
- ^ Levine SP, Huggins JE, BeMent SL, Kushwaha RK, Schuh LA, Rohde MM, et al. (June 2000). "A direct brain interface based on event-related potentials". IEEE Transactions on Rehabilitation Engineering. 8 (2): 180–185. doi:10.1109/86.847809. PMID 10896180.
- ^ Bird JJ, Manso LJ, Ribeiro EP, Ekárt A, Faria DR (September 2018). A Study on Mental State Classification using EEG-based Brain-Machine Interface. Madeira Island, Portugal: 9th international Conference on Intelligent Systems 2018. Retrieved 3 December 2018.
- ^ Bird JJ, Ekart A, Buckingham CD, Faria DR (2019). Mental Emotional Sentiment Classification with an EEG-based Brain-Machine Interface. St Hugh's College, University of Oxford, United Kingdom: The International Conference on Digital Image and Signal Processing (DISP'19). Archived from the original on 3 December 2018. Retrieved 3 December 2018.
- ^ Vanneste S, Song JJ, De Ridder D (March 2018). "Thalamocortical dysrhythmia detected by machine learning". Nature Communications. 9 (1): 1103. Bibcode:2018NatCo...9.1103V. doi:10.1038/s41467-018-02820-0. PMC 5856824. PMID 29549239.
- ^ Straebel V, Thoben W (2014). "Alvin Lucier's music for solo performer: experimental music beyond sonification". Organised Sound. 19 (1): 17–29. doi:10.1017/S135577181300037X. S2CID 62506825.
- ^ a b Wolpaw, J.R. and Wolpaw, E.W. (2012). "Brain-Computer Interfaces: Something New Under the Sun". In: Brain-Computer Interfaces: Principles and Practice, Wolpaw, J.R. and Wolpaw (eds.), E.W. Oxford University Press.
- ^ Wolpaw JR, Birbaumer N, McFarland DJ, Pfurtscheller G, Vaughan TM (June 2002). "Brain-computer interfaces for communication and control". Clinical Neurophysiology. 113 (6): 767–791. doi:10.1016/s1388-2457(02)00057-3. PMID 12048038. S2CID 17571592.
- ^ Allison BZ, Wolpaw EW, Wolpaw JR (July 2007). "Brain-computer interface systems: progress and prospects". Expert Review of Medical Devices. 4 (4): 463–474. doi:10.1586/17434440.4.4.463. PMID 17605682. S2CID 4690450.
- ^ a b Bozinovski S, Bozinovska L (2019). "Brain-computer interface in Europe: The thirtieth anniversary". Automatika. 60 (1): 36–47. doi:10.1080/00051144.2019.1570644.
- ^ Vidal, Jacques J. (1977). "Real-time detection of brain events in EEG" (PDF). Proceedings of the IEEE. 65 (5): 633–641. doi:10.1109/PROC.1977.10542. S2CID 7928242. Archived from the original (PDF) on 19 July 2015. Retrieved 4 November 2022.
- ^ S. Bozinovski, M. Sestakov, L. Bozinovska: Using EEG alpha rhythm to control a mobile robot, In G. Harris, C. Walker (eds.) Proc. IEEE Annual Conference of Medical and Biological Society, p. 1515-1516, New Orleans, 1988
- ^ S. Bozinovski: Mobile robot trajectory control: From fixed rails to direct bioelectric control, In O. Kaynak (ed.) Proc. IEEE Workshop on Intelligent Motion Control, p. 63-67, Istanbul, 1990
- ^ M. Lebedev: Augmentation of sensorimotor functions with neural prostheses. Opera Medica and Physiologica. Vol. 2 (3): 211-227, 2016
- ^ M. Lebedev, M. Nicolelis: Brain-machine interfaces: from basic science to neuroprostheses and neurorehabilitation, Physiological Review 97:737-867, 2017
- ^ L. Bozinovska, G. Stojanov, M. Sestakov, S. Bozinovski: CNV pattern recognition: step toward a cognitive wave observation, In L. Torres, E. Masgrau, E. Lagunas (eds.) Signal Processing V: Theories and Applications, Proc. EUSIPCO-90: Fifth European Signal Processing Conference, Elsevier, p. 1659-1662, Barcelona, 1990
- ^ L. Bozinovska, S. Bozinovski, G. Stojanov, Electroexpectogram: experimental design and algorithms, In Proc IEEE International Biomedical Engineering Days, p. 55-60, Istanbul, 1992
- ^ Miranda RA, Casebeer WD, Hein AM, Judy JW, Krotkov EP, Laabs TL, et al. (April 2015). "DARPA-funded efforts in the development of novel brain-computer interface technologies". Journal of Neuroscience Methods. 244: 52–67. doi:10.1016/j.jneumeth.2014.07.019. PMID 25107852. S2CID 14678623.
- ^ Jacobs M, Premji A, Nelson AJ (16 May 2012). "Plasticity-inducing TMS protocols to investigate somatosensory control of hand function". Neural Plasticity. 2012: 350574. doi:10.1155/2012/350574. PMC 3362131. PMID 22666612.
- ^ Fox, Maggie (October 13, 2016). "Brain Chip Helps Paralyzed Man Feel His Fingers". NBC News. Retrieved 23 March 2021.
- ^ Hatmaker, Taylor (July 10, 2017). "DARPA awards $65 million to develop the perfect, tiny two-way brain-computer inerface". Tech Crunch. Retrieved 23 March 2021.
- ^ Stacey, Kevin (July 10, 2017). "Brown to receive up to $19M to engineer next-generation brain-computer interface". Brown University. Retrieved 23 March 2021.
- ^ "Minimally Invasive "Stentrode" Shows Potential as Neural Interface for Brain". Defense Advanced Research Projects Agency (DARPA). 2016-02-08. Retrieved 23 March 2021.
- ^ "Cochlear Implants". National Institute on Deafness and Other Communication Disorders. February 2016. Retrieved 1 April 2024.
- ^ Miguel Nicolelis et al. (2001) Duke neurobiologist has developed system that allows monkeys to control robot arms via brain signals Archived 19 December 2008 at the Wayback Machine
- ^ Baum M (6 September 2008). "Monkey Uses Brain Power to Feed Itself With Robotic Arm". Pitt Chronicle. Archived from the original on 10 September 2009. Retrieved 6 July 2009.
- ^ Lewis T (November 2020). "Elon Musk's Pig-Brain Implant Is Still a Long Way from 'Solving Paralysis'". Scientific American. Retrieved 23 March 2021.
- ^ Shead S (February 2021). "Elon Musk says his start-up Neuralink has wired up a monkey to play video games using its mind". CNBC. Retrieved 23 March 2021.
- ^ Fetz EE (February 1969). "Operant conditioning of cortical unit activity". Science. 163 (3870): 955–958. Bibcode:1969Sci...163..955F. doi:10.1126/science.163.3870.955. PMID 4974291. S2CID 45427819.
- ^ Schmidt EM, McIntosh JS, Durelli L, Bak MJ (September 1978). "Fine control of operantly conditioned firing patterns of cortical neurons". Experimental Neurology. 61 (2): 349–369. doi:10.1016/0014-4886(78)90252-2. PMID 101388. S2CID 37539476.
- ^ Georgopoulos AP, Lurito JT, Petrides M, Schwartz AB, Massey JT (January 1989). "Mental rotation of the neuronal population vector". Science. 243 (4888): 234–236. Bibcode:1989Sci...243..234G. doi:10.1126/science.2911737. PMID 2911737. S2CID 37161168.
- ^ Lebedev MA, Nicolelis MA (September 2006). "Brain-machine interfaces: past, present and future". Trends in Neurosciences. 29 (9): 536–546. doi:10.1016/j.tins.2006.07.004. PMID 16859758. S2CID 701524.
- ^ Stanley GB, Li FF, Dan Y (September 1999). "Reconstruction of natural scenes from ensemble responses in the lateral geniculate nucleus". The Journal of Neuroscience. 19 (18): 8036–8042. doi:10.1523/JNEUROSCI.19-18-08036.1999. PMC 6782475. PMID 10479703.
- ^ Wessberg J, Stambaugh CR, Kralik JD, Beck PD, Laubach M, Chapin JK, et al. (November 2000). "Real-time prediction of hand trajectory by ensembles of cortical neurons in primates". Nature. 408 (6810): 361–365. Bibcode:2000Natur.408..361W. doi:10.1038/35042582. PMID 11099043. S2CID 795720.
- ^ a b Carmena JM, Lebedev MA, Crist RE, O'Doherty JE, Santucci DM, Dimitrov DF, et al. (November 2003). "Learning to control a brain-machine interface for reaching and grasping by primates". PLOS Biology. 1 (2): E42. doi:10.1371/journal.pbio.0000042. PMC 261882. PMID 14624244.
- ^ a b Lebedev MA, Carmena JM, O'Doherty JE, Zacksenhouse M, Henriquez CS, Principe JC, Nicolelis MA (May 2005). "Cortical ensemble adaptation to represent velocity of an artificial actuator controlled by a brain-machine interface". The Journal of Neuroscience. 25 (19): 4681–4693. doi:10.1523/JNEUROSCI.4088-04.2005. PMC 6724781. PMID 15888644.
- ^ O'Doherty JE, Lebedev MA, Ifft PJ, Zhuang KZ, Shokur S, Bleuler H, Nicolelis MA (October 2011). "Active tactile exploration using a brain-machine-brain interface". Nature. 479 (7372): 228–231. Bibcode:2011Natur.479..228O. doi:10.1038/nature10489. PMC 3236080. PMID 21976021.
- ^ Serruya MD, Hatsopoulos NG, Paninski L, Fellows MR, Donoghue JP (March 2002). "Instant neural control of a movement signal". Nature. 416 (6877): 141–142. Bibcode:2002Natur.416..141S. doi:10.1038/416141a. PMID 11894084. S2CID 4383116.
- ^ Taylor DM, Tillery SI, Schwartz AB (June 2002). "Direct cortical control of 3D neuroprosthetic devices". Science. 296 (5574): 1829–1832. Bibcode:2002Sci...296.1829T. CiteSeerX 10.1.1.1027.4335. doi:10.1126/science.1070291. PMID 12052948. S2CID 9402759.
- ^ Pitt team to build on brain-controlled arm Archived 4 July 2007 at the Wayback Machine, Pittsburgh Tribune Review, 5 September 2006.
- ^ Video on YouTube
- ^ Velliste M, Perel S, Spalding MC, Whitford AS, Schwartz AB (June 2008). "Cortical control of a prosthetic arm for self-feeding". Nature. 453 (7198): 1098–1101. Bibcode:2008Natur.453.1098V. doi:10.1038/nature06996. PMID 18509337. S2CID 4404323.
- ^ Musallam S, Corneil BD, Greger B, Scherberger H, Andersen RA (July 2004). "Cognitive control signals for neural prosthetics". Science. 305 (5681): 258–262. Bibcode:2004Sci...305..258M. doi:10.1126/science.1097938. PMID 15247483. S2CID 3112034.
- ^ Santucci DM, Kralik JD, Lebedev MA, Nicolelis MA (September 2005). "Frontal and parietal cortical ensembles predict single-trial muscle activity during reaching movements in primates". The European Journal of Neuroscience. 22 (6): 1529–1540. doi:10.1111/j.1460-9568.2005.04320.x. PMID 16190906. S2CID 31277881.
- ^ Anumanchipalli GK, Chartier J, Chang EF (April 2019). "Speech synthesis from neural decoding of spoken sentences". Nature. 568 (7753): 493–498. Bibcode:2019Natur.568..493A. doi:10.1038/s41586-019-1119-1. PMC 9714519. PMID 31019317. S2CID 129946122.
- ^ Pandarinath C, Ali YH (April 2019). "Brain implants that let you speak your mind". Nature. 568 (7753): 466–467. Bibcode:2019Natur.568..466P. doi:10.1038/d41586-019-01181-y. PMID 31019323.
- ^ a b Moses DA, Metzger SL, Liu JR, Anumanchipalli GK, Makin JG, Sun PF, et al. (July 2021). "Neuroprosthesis for Decoding Speech in a Paralyzed Person with Anarthria". The New England Journal of Medicine. 385 (3): 217–227. doi:10.1056/NEJMoa2027540. PMC 8972947. PMID 34260835. S2CID 235907121.
- ^ Belluck, Pam (14 July 2021). "Tapping Into the Brain to Help a Paralyzed Man Speak". The New York Times.
- ^ "Using BCI2000 in BCI Research". National Center for Adaptive Neurotechnology. Retrieved 5 December 2023.
- ^ Huber D, Petreanu L, Ghitani N, Ranade S, Hromádka T, Mainen Z, Svoboda K (January 2008). "Sparse optical microstimulation in barrel cortex drives learned behaviour in freely moving mice". Nature. 451 (7174): 61–64. Bibcode:2008Natur.451...61H. doi:10.1038/nature06445. PMC 3425380. PMID 18094685.
- ^ Nicolelis MA, Lebedev MA (July 2009). "Principles of neural ensemble physiology underlying the operation of brain-machine interfaces". Nature Reviews. Neuroscience. 10 (7): 530–540. doi:10.1038/nrn2653. PMID 19543222. S2CID 9290258.
- ^ a b Zander TO, Kothe C (April 2011). "Towards passive brain-computer interfaces: applying brain-computer interface technology to human-machine systems in general". Journal of Neural Engineering. 8 (2): 025005. Bibcode:2011JNEng...8b5005Z. doi:10.1088/1741-2560/8/2/025005. PMID 21436512. S2CID 37168897.
- ^ Richardson AG, Ghenbot Y, Liu X, Hao H, Rinehart C, DeLuccia S, et al. (August 2019). "Learning active sensing strategies using a sensory brain-machine interface". Proceedings of the National Academy of Sciences of the United States of America. 116 (35): 17509–17514. Bibcode:2019PNAS..11617509R. doi:10.1073/pnas.1909953116. PMC 6717311. PMID 31409713.
- ^ Abdulkader SN, Atia A, Mostafa MS (July 2015). "Brain computer interfacing: Applications and challenges". Egyptian Informatics Journal. 16 (2): 213–230. doi:10.1016/j.eij.2015.06.002. ISSN 1110-8665.
- ^ Polikov VS, Tresco PA, Reichert WM (October 2005). "Response of brain tissue to chronically implanted neural electrodes". Journal of Neuroscience Methods. 148 (1): 1–18. doi:10.1016/j.jneumeth.2005.08.015. PMID 16198003. S2CID 11248506.
- ^ "Vision quest". Wired. (September 2002).
- ^ Kotler S. "Vision Quest". Wired. ISSN 1059-1028. Retrieved 2021-11-10.
- ^ Tuller D (1 November 2004). "Dr. William Dobelle, Artificial Vision Pioneer, Dies at 62". The New York Times.
- ^ Naumann J (2012). Search for Paradise: A Patient's Account of the Artificial Vision Experiment. Xlibris. ISBN 978-1-4797-0920-5.
- ^ nurun.com (28 November 2012). "Mr. Jen Naumann's high-tech paradise lost". Thewhig.com. Retrieved 19 December 2016.
- ^ Kennedy PR, Bakay RA (June 1998). "Restoration of neural output from a paralyzed patient by a direct brain connection". NeuroReport. 9 (8): 1707–1711. doi:10.1097/00001756-199806010-00007. PMID 9665587. S2CID 5681602.
- ^ Hochberg LR, Serruya MD, Friehs GM, Mukand JA, Saleh M, Caplan AH, et al. (July 2006). "Neuronal ensemble control of prosthetic devices by a human with tetraplegia". Nature. 442 (7099). Gerhard M. Friehs, Jon A. Mukand, Maryam Saleh, Abraham H. Caplan, Almut Branner, David Chen, Richard D. Penn and John P. Donoghue: 164–171. Bibcode:2006Natur.442..164H. doi:10.1038/nature04970. PMID 16838014. S2CID 4347367.
- ^ Martins Iduwe. "Brain Computer Interface". Academia.edu. Retrieved 5 December 2023.
- ^ Hochberg LR, Bacher D, Jarosiewicz B, Masse NY, Simeral JD, Vogel J, et al. (May 2012). "Reach and grasp by people with tetraplegia using a neurally controlled robotic arm". Nature. 485 (7398): 372–375. Bibcode:2012Natur.485..372H. doi:10.1038/nature11076. PMC 3640850. PMID 22596161.
- ^ Collinger JL, Wodlinger B, Downey JE, Wang W, Tyler-Kabara EC, Weber DJ, et al. (February 2013). "High-performance neuroprosthetic control by an individual with tetraplegia". Lancet. 381 (9866): 557–564. doi:10.1016/S0140-6736(12)61816-9. PMC 3641862. PMID 23253623.
- ^ Willett FR, Avansino DT, Hochberg LR, Henderson JM, Shenoy KV (May 2021). "High-performance brain-to-text communication via handwriting". Nature. 593 (7858): 249–254. Bibcode:2021Natur.593..249W. doi:10.1038/s41586-021-03506-2. PMC 8163299. PMID 33981047.
- ^ Willett FR (2021). "A High-Performance Handwriting BCI". In Guger C, Allison BZ, Gunduz A (eds.). Brain-Computer Interface Research: A State-of-the-Art Summary 10. SpringerBriefs in Electrical and Computer Engineering. Cham: Springer International Publishing. pp. 105–109. doi:10.1007/978-3-030-79287-9_11. ISBN 978-3-030-79287-9. S2CID 239736609.
- ^ Hamliton J (14 July 2021). "Experimental Brain Implant Lets Man With Paralysis Turn His Thoughts Into Words". All Things Considered. NPR.
- ^ Pandarinath C, Bensmaia SJ (September 2021). "The science and engineering behind sensitized brain-controlled bionic hands". Physiological Reviews. 102 (2): 551–604. doi:10.1152/physrev.00034.2020. PMC 8742729. PMID 34541898. S2CID 237574228.
- ^ Willett, Francis R.; Kunz, Erin M.; Fan, Chaofei; Avansino, Donald T.; Wilson, Guy H.; Choi, Eun Young; Kamdar, Foram; Glasser, Matthew F.; Hochberg, Leigh R.; Druckmann, Shaul; Shenoy, Krishna V.; Henderson, Jaimie M. (2023-08-23). "A high-performance speech neuroprosthesis". Nature. 620 (7976): 1031–1036. Bibcode:2023Natur.620.1031W. doi:10.1038/s41586-023-06377-x. ISSN 1476-4687. PMC 10468393. PMID 37612500.
- ^ Metzger, Sean L.; Littlejohn, Kaylo T.; Silva, Alexander B.; Moses, David A.; Seaton, Margaret P.; Wang, Ran; Dougherty, Maximilian E.; Liu, Jessie R.; Wu, Peter; Berger, Michael A.; Zhuravleva, Inga; Tu-Chan, Adelyn; Ganguly, Karunesh; Anumanchipalli, Gopala K.; Chang, Edward F. (2023-08-23). "A high-performance neuroprosthesis for speech decoding and avatar control". Nature. 620 (7976): 1037–1046. Bibcode:2023Natur.620.1037M. doi:10.1038/s41586-023-06443-4. ISSN 1476-4687. PMC 10826467. PMID 37612505. S2CID 261098775.
- ^ Naddaf, Miryam (2023-08-23). "Brain-reading devices allow paralysed people to talk using their thoughts". Nature. 620 (7976): 930–931. Bibcode:2023Natur.620..930N. doi:10.1038/d41586-023-02682-7. PMID 37612493. S2CID 261099321.
- ^ Zhang M, Tang Z, Liu X, Van der Spiegel J (April 2020). "Electronic neural interfaces". Nature Electronics. 3 (4): 191–200. doi:10.1038/s41928-020-0390-3. ISSN 2520-1131. S2CID 216508360.
- ^ Hodgkin AL, Huxley AF (August 1952). "A quantitative description of membrane current and its application to conduction and excitation in nerve". The Journal of Physiology. 117 (4): 500–544. doi:10.1113/jphysiol.1952.sp004764. PMC 1392413. PMID 12991237.
- ^ a b Obien ME, Deligkaris K, Bullmann T, Bakkum DJ, Frey U (2015). "Revealing neuronal function through microelectrode array recordings". Frontiers in Neuroscience. 8: 423. doi:10.3389/fnins.2014.00423. PMC 4285113. PMID 25610364.
- ^ a b Harrison RR (July 2008). "The Design of Integrated Circuits to Observe Brain Activity". Proceedings of the IEEE. 96 (7): 1203–1216. doi:10.1109/JPROC.2008.922581. ISSN 1558-2256. S2CID 7020369.
- ^ Haci D, Liu Y, Ghoreishizadeh SS, Constandinou TG (February 2020). "Key Considerations for Power Management in Active Implantable Medical Devices". 2020 IEEE 11th Latin American Symposium on Circuits & Systems (LASCAS). pp. 1–4. doi:10.1109/LASCAS45839.2020.9069004. ISBN 978-1-7281-3427-7. S2CID 215817530.
- ^ Downey JE, Schwed N, Chase SM, Schwartz AB, Collinger JL (August 2018). "Intracortical recording stability in human brain-computer interface users". Journal of Neural Engineering. 15 (4): 046016. Bibcode:2018JNEng..15d6016D. doi:10.1088/1741-2552/aab7a0. PMID 29553484. S2CID 3961913.
- ^ Freire MA, Morya E, Faber J, Santos JR, Guimaraes JS, Lemos NA, Sameshima K, Pereira A, Ribeiro S, Nicolelis M (November 2011). "Comprehensive analysis of tissue preservation and recording quality from chronic multielectrode implants". PLOS ONE. 6 (11): e27554. Bibcode:2011PLoSO...627554F. doi:10.1371/journal.pone.0027554. PMC 4476592. PMID 26098896.
- ^ Szostak KM, Grand L, Constandinou TG (2017). "Neural Interfaces for Intracortical Recording: Requirements, Fabrication Methods, and Characteristics". Frontiers in Neuroscience. 11: 665. doi:10.3389/fnins.2017.00665. PMC 5725438. PMID 29270103.
- ^ a b Saxena T, Karumbaiah L, Gaupp EA, Patkar R, Patil K, Betancur M, et al. (July 2013). "The impact of chronic blood-brain barrier breach on intracortical electrode function". Biomaterials. 34 (20): 4703–4713. doi:10.1016/j.biomaterials.2013.03.007. PMID 23562053.
- ^ Nolta NF, Christensen MB, Crane PD, Skousen JL, Tresco PA (2015-06-01). "BBB leakage, astrogliosis, and tissue loss correlate with silicon microelectrode array recording performance". Biomaterials. 53: 753–762. doi:10.1016/j.biomaterials.2015.02.081. PMID 25890770.
- ^ Robinson JT, Pohlmeyer E, Gather MC, Kemere C, Kitching JE, Malliaras GG, et al. (November 2019). "Developing Next-generation Brain Sensing Technologies - A Review". IEEE Sensors Journal. 19 (22): 10163–10175. doi:10.1109/JSEN.2019.2931159. PMC 7047830. PMID 32116472.
- ^ Luan L, Wei X, Zhao Z, Siegel JJ, Potnis O, Tuppen CA, et al. (February 2017). "Ultraflexible nanoelectronic probes form reliable, glial scar-free neural integration". Science Advances. 3 (2): e1601966. Bibcode:2017SciA....3E1966L. doi:10.1126/sciadv.1601966. PMC 5310823. PMID 28246640.
- ^ Frank JA, Antonini MJ, Anikeeva P (September 2019). "Next-generation interfaces for studying neural function". Nature Biotechnology. 37 (9): 1013–1023. doi:10.1038/s41587-019-0198-8. PMC 7243676. PMID 31406326.
- ^ a b Hong G, Viveros RD, Zwang TJ, Yang X, Lieber CM (July 2018). "Tissue-like Neural Probes for Understanding and Modulating the Brain". Biochemistry. 57 (27): 3995–4004. doi:10.1021/acs.biochem.8b00122. PMC 6039269. PMID 29529359.
- ^ Viveros RD, Zhou T, Hong G, Fu TM, Lin HG, Lieber CM (June 2019). "Advanced One- and Two-Dimensional Mesh Designs for Injectable Electronics". Nano Letters. 19 (6): 4180–4187. Bibcode:2019NanoL..19.4180V. doi:10.1021/acs.nanolett.9b01727. PMC 6565464. PMID 31075202.
- ^ Gulati T, Won SJ, Ramanathan DS, Wong CC, Bodepudi A, Swanson RA, Ganguly K (June 2015). "Robust neuroprosthetic control from the stroke perilesional cortex". The Journal of Neuroscience. 35 (22): 8653–8661. doi:10.1523/JNEUROSCI.5007-14.2015. PMC 6605327. PMID 26041930.
- ^ Soldozy S, Young S, Kumar JS, Capek S, Felbaum DR, Jean WC, et al. (July 2020). "A systematic review of endovascular stent-electrode arrays, a minimally invasive approach to brain-machine interfaces". Neurosurgical Focus. 49 (1): E3. doi:10.3171/2020.4.FOCUS20186. PMID 32610291. S2CID 220308983.
- ^ a b Opie N (2021). "The StentrodeTM Neural Interface System". In Guger C, Allison BZ, Tangermann M (eds.). Brain-Computer Interface Research. SpringerBriefs in Electrical and Computer Engineering. Cham: Springer International Publishing. pp. 127–132. doi:10.1007/978-3-030-60460-8_13. ISBN 978-3-030-60460-8. S2CID 234102889.
- ^ Teleb MS, Cziep ME, Lazzaro MA, Gheith A, Asif K, Remler B, Zaidat OO (May 2014). "Idiopathic Intracranial Hypertension. A Systematic Analysis of Transverse Sinus Stenting". Interventional Neurology. 2 (3): 132–143. doi:10.1159/000357503. PMC 4080637. PMID 24999351.
- ^ Bryson S (5 November 2020). "Stentrode Device Allows Computer Control by ALS Patients with Partial Upper Limb Paralysis". ALS News Today.
- ^ Lanese, Nicoletta (12 January 2023). "New 'thought-controlled' device reads brain activity through the jugular". livescience.com. Archived from the original on 16 February 2023. Retrieved 16 February 2023.
- ^ Mitchell, Peter; Lee, Sarah C. M.; Yoo, Peter E.; Morokoff, Andrew; Sharma, Rahul P.; Williams, Daryl L.; MacIsaac, Christopher; Howard, Mark E.; Irving, Lou; Vrljic, Ivan; Williams, Cameron; Bush, Steven; Balabanski, Anna H.; Drummond, Katharine J.; Desmond, Patricia; Weber, Douglas; Denison, Timothy; Mathers, Susan; O’Brien, Terence J.; Mocco, J.; Grayden, David B.; Liebeskind, David S.; Opie, Nicholas L.; Oxley, Thomas J.; Campbell, Bruce C. V. (9 January 2023). "Assessment of Safety of a Fully Implanted Endovascular Brain-Computer Interface for Severe Paralysis in 4 Patients: The Stentrode With Thought-Controlled Digital Switch (SWITCH) Study". JAMA Neurology. 80 (3): 270–278. doi:10.1001/jamaneurol.2022.4847. ISSN 2168-6149. PMC 9857731. PMID 36622685. S2CID 255545643.
- ^ Serruya M, Donoghue J (2004). "Chapter III: Design Principles of a Neuromotor Prosthetic Device" (PDF). In Horch KW, Dhillon GS (eds.). Neuroprosthetics: Theory and Practice. Imperial College Press. pp. 1158–1196. doi:10.1142/9789812561763_0040. Archived from the original (PDF) on 4 April 2005.
- ^ "Teenager moves video icons just by imagination". Press release. Washington University in St Louis. 9 October 2006.
- ^ Schalk G, Miller KJ, Anderson NR, Wilson JA, Smyth MD, Ojemann JG, et al. (March 2008). "Two-dimensional movement control using electrocorticographic signals in humans". Journal of Neural Engineering. 5 (1): 75–84. Bibcode:2008JNEng...5...75S. doi:10.1088/1741-2560/5/1/008. PMC 2744037. PMID 18310813.
- ^ Nicolas-Alonso LF, Gomez-Gil J (2012-01-31). "Brain computer interfaces, a review". Sensors. 12 (2): 1211–1279. Bibcode:2012Senso..12.1211N. doi:10.3390/s120201211. PMC 3304110. PMID 22438708.
- ^ Yanagisawa T (2011). "Electrocorticographic Control of Prosthetic Arm in Paralyzed Patients". American Neurological Association. Vol. 71, no. 3. pp. 353–361. doi:10.1002/ana.22613.
ECoG- Based BCI has advantage in signal and durability that are absolutely necessary for clinical application
- ^ a b Pei X (2011). "Decoding Vowels and Consonants in Spoken and Imagined Words Using Electrocorticographic Signals in Humans". J Neural Eng 046028th ser. 8.4. PMID 21750369.
Justin Williams, a biomedical engineer at the university, has already transformed the ECoG implant into a micro device that can be installed with a minimum of fuss. It has been tested in animals for a long period of time – the micro ECoG stays in place and doesn't seem to negatively affect the immune system.
- ^ Makin JG, Moses DA, Chang EF (2021). "Speech Decoding as Machine Translation". In Guger C, Allison BZ, Gunduz A (eds.). Brain-Computer Interface Research. SpringerBriefs in Electrical and Computer Engineering. Cham: Springer International Publishing. pp. 23–33. doi:10.1007/978-3-030-79287-9_3. ISBN 978-3-030-79287-9. S2CID 239756345.
- ^ Makin JG, Moses DA, Chang EF (April 2020). "Machine translation of cortical activity to text with an encoder-decoder framework". Nature Neuroscience. 23 (4): 575–582. doi:10.1038/s41593-020-0608-8. PMC 10560395. PMID 32231340. S2CID 214704481.
- ^ Gallegos-Ayala G, Furdea A, Takano K, Ruf CA, Flor H, Birbaumer N (May 2014). "Brain communication in a completely locked-in patient using bedside near-infrared spectroscopy". Neurology. 82 (21): 1930–1932. doi:10.1212/WNL.0000000000000449. PMC 4049706. PMID 24789862.
- ^ Chaudhary U, Xia B, Silvoni S, Cohen LG, Birbaumer N (January 2017). "Brain-Computer Interface-Based Communication in the Completely Locked-In State". PLOS Biology. 15 (1): e1002593. doi:10.1371/journal.pbio.1002593. PMC 5283652. PMID 28141803.
- ^ Winters, Jeffrey (May 2003). "Communicating by Brain Waves". Psychology Today.
- ^ Adrijan Bozinovski "CNV flip-flop as a brain-computer interface paradigm" In J. Kern, S. Tonkovic, et al. (Eds) Proc 7th Conference of the Croatian Association of Medical Informatics, pp. 149-154, Rijeka, 2005
- ^ Bozinovski, Adrijan; Bozinovska, Liljana (2009). Anticipatory brain potentials in a Brain-Robot Interface paradigm. 2009 4th International IEEE/EMBS Conference on Neural Engineering. IEEE. pp. 451–454. doi:10.1109/ner.2009.5109330.
- ^ Božinovski, Adrijan; Tonković, Stanko; Išgum, Velimir; Božinovska, Liljana (2011). "Robot Control Using Anticipatory Brain Potentials". Automatika. 52 (1): 20–30. doi:10.1080/00051144.2011.11828400. S2CID 33223634.
- ^ Bozinovski, Stevo; Bozinovski, Adrijan (2015). "Mental States, EEG Manifestations, and Mentally Emulated Digital Circuits for Brain-Robot Interaction". IEEE Transactions on Autonomous Mental Development. 7 (1). Institute of Electrical and Electronics Engineers (IEEE): 39–51. doi:10.1109/tamd.2014.2387271. ISSN 1943-0604. S2CID 21464338.
- ^ Yuan H, Liu T, Szarkowski R, Rios C, Ashe J, He B (February 2010). "Negative covariation between task-related responses in alpha/beta-band activity and BOLD in human sensorimotor cortex: an EEG and fMRI study of motor imagery and movements". NeuroImage. 49 (3): 2596–2606. doi:10.1016/j.neuroimage.2009.10.028. PMC 2818527. PMID 19850134.
- ^ Doud AJ, Lucas JP, Pisansky MT, He B (2011). Gribble PL (ed.). "Continuous three-dimensional control of a virtual helicopter using a motor imagery based brain-computer interface". PLOS ONE. 6 (10): e26322. Bibcode:2011PLoSO...626322D. doi:10.1371/journal.pone.0026322. PMC 3202533. PMID 22046274.
- ^ "Thought-guided helicopter takes off". BBC News. 5 June 2013. Retrieved 5 June 2013.
- ^ Qin L, Ding L, He B (September 2004). "Motor imagery classification by means of source analysis for brain-computer interface applications". Journal of Neural Engineering. 1 (3): 135–141. Bibcode:2004JNEng...1..135Q. doi:10.1088/1741-2560/1/3/002. PMC 1945182. PMID 15876632.
- ^ Höhne J, Holz E, Staiger-Sälzer P, Müller KR, Kübler A, Tangermann M (2014). "Motor imagery for severely motor-impaired patients: evidence for brain-computer interfacing as superior control solution". PLOS ONE. 9 (8): e104854. Bibcode:2014PLoSO...9j4854H. doi:10.1371/journal.pone.0104854. PMC 4146550. PMID 25162231.
- ^ Maskeliunas R, Damasevicius R, Martisius I, Vasiljevas M (2016). "Consumer-grade EEG devices: are they usable for control tasks?". PeerJ. 4: e1746. doi:10.7717/peerj.1746. PMC 4806709. PMID 27014511.
- ^ Bird JJ, Faria DR, Manso LJ, Ekárt A, Buckingham CD (2019-03-13). "A Deep Evolutionary Approach to Bioinspired Classifier Optimisation for Brain-Machine Interaction". Complexity. 2019. Hindawi Limited: 1–14. arXiv:1908.04784. doi:10.1155/2019/4316548. ISSN 1076-2787.
- ^ Mansour S, Ang KK, Nair KP, Phua KS, Arvaneh M (January 2022). "Efficacy of Brain-Computer Interface and the Impact of Its Design Characteristics on Poststroke Upper-limb Rehabilitation: A Systematic Review and Meta-analysis of Randomized Controlled Trials". Clinical EEG and Neuroscience. 53 (1): 79–90. doi:10.1177/15500594211009065. PMC 8619716. PMID 33913351. S2CID 233446181.
- ^ Baniqued PD, Stanyer EC, Awais M, Alazmani A, Jackson AE, Mon-Williams MA, et al. (January 2021). "Brain-computer interface robotics for hand rehabilitation after stroke: a systematic review". Journal of Neuroengineering and Rehabilitation. 18 (1): 15. doi:10.1186/s12984-021-00820-8. PMC 7825186. PMID 33485365.
- ^ Taheri BA, Knight RT, Smith RL (May 1994). "A dry electrode for EEG recording". Electroencephalography and Clinical Neurophysiology. 90 (5): 376–383. doi:10.1016/0013-4694(94)90053-1. PMID 7514984.
- ^ Alizadeh-Taheri B (1994). Active Micromachined Scalp Electrode Array for Eeg Signal Recording (PHD Thesis thesis). p. 82. Bibcode:1994PhDT........82A.
- ^ Hockenberry, John (August 2001). "The Next Brainiacs". Wired. Vol. 9, no. 8.
- ^ Lin CT, Ko LW, Chang CJ, Wang YT, Chung CH, Yang FS, et al. (2009), "Wearable and Wireless Brain-Computer Interface and Its Applications", Foundations of Augmented Cognition. Neuroergonomics and Operational Neuroscience, Lecture Notes in Computer Science, vol. 5638, Springer Berlin Heidelberg, pp. 741–748, doi:10.1007/978-3-642-02812-0_84, ISBN 978-3-642-02811-3, S2CID 14515754
- ^ a b c d e Wang YT, Wang Y, Jung TP (April 2011). "A cell-phone-based brain-computer interface for communication in daily life". Journal of Neural Engineering. 8 (2): 025018. Bibcode:2011JNEng...8b5018W. doi:10.1088/1741-2560/8/2/025018. PMID 21436517. S2CID 10943518.
- ^ Guger C, Allison BZ, Großwindhager B, Prückl R, Hintermüller C, Kapeller C, et al. (2012). "How Many People Could Use an SSVEP BCI?". Frontiers in Neuroscience. 6: 169. doi:10.3389/fnins.2012.00169. PMC 3500831. PMID 23181009.
- ^ a b Lin YP, Wang Y, Jung TP (2013). "A mobile SSVEP-based brain-computer interface for freely moving humans: The robustness of canonical correlation analysis to motion artifacts". 2013 35th Annual International Conference of the IEEE Engineering in Medicine and Biology Society (EMBC). Vol. 2013. pp. 1350–1353. doi:10.1109/EMBC.2013.6609759. ISBN 978-1-4577-0216-7. PMID 24109946. S2CID 23136360.
- ^ Rashid M, Sulaiman N, Abdul Majeed AP, Musa RM, Ab Nasir AF, Bari BS, Khatun S (2020). "Current Status, Challenges, and Possible Solutions of EEG-Based Brain-Computer Interface: A Comprehensive Review". Frontiers in Neurorobotics. 14: 25. doi:10.3389/fnbot.2020.00025. PMC 7283463. PMID 32581758.
- ^ US 20130127708, issued 23 May 2013
- ^ a b c Wang YT, Wang Y, Cheng CK, Jung TP (2013). "Developing stimulus presentation on mobile devices for a truly portable SSVEP-based BCI". 2013 35th Annual International Conference of the IEEE Engineering in Medicine and Biology Society (EMBC). Vol. 2013. pp. 5271–5274. doi:10.1109/EMBC.2013.6610738. ISBN 978-1-4577-0216-7. PMID 24110925. S2CID 14324159.
- ^ Bin G, Gao X, Yan Z, Hong B, Gao S (August 2009). "An online multi-channel SSVEP-based brain-computer interface using a canonical correlation analysis method". Journal of Neural Engineering. 6 (4): 046002. Bibcode:2009JNEng...6d6002B. doi:10.1088/1741-2560/6/4/046002. PMID 19494422. S2CID 32640699.
- ^ Symeonidou ER, Nordin AD, Hairston WD, Ferris DP (April 2018). "Effects of Cable Sway, Electrode Surface Area, and Electrode Mass on Electroencephalography Signal Quality during Motion". Sensors. 18 (4): 1073. Bibcode:2018Senso..18.1073S. doi:10.3390/s18041073. PMC 5948545. PMID 29614020.
- ^ Wang Y, Wang R, Gao X, Hong B, Gao S (June 2006). "A practical VEP-based brain-computer interface". IEEE Transactions on Neural Systems and Rehabilitation Engineering. 14 (2): 234–239. doi:10.1109/TNSRE.2006.875576. PMID 16792302.
- ^ Pfurtscheller G, Müller GR, Pfurtscheller J, Gerner HJ, Rupp R (November 2003). "'Thought'--control of functional electrical stimulation to restore hand grasp in a patient with tetraplegia". Neuroscience Letters. 351 (1): 33–36. doi:10.1016/S0304-3940(03)00947-9. PMID 14550907. S2CID 38568963.
- ^ Do AH, Wang PT, King CE, Chun SN, Nenadic Z (December 2013). "Brain-computer interface controlled robotic gait orthosis". Journal of Neuroengineering and Rehabilitation. 10 (1): 111. doi:10.1186/1743-0003-10-111. PMC 3907014. PMID 24321081.
- ^ Subject with Paraplegia Operates BCI-controlled RoGO (4x) at YouTube.com
- ^ Alex Blainey controls a cheap consumer robot arm using the EPOC headset via a serial relay port at YouTube.com
- ^ Drummond, Katie (14 May 2009). "Pentagon Preps Soldier Telepathy Push". Wired. Retrieved 6 May 2009.
- ^ Ranganatha Sitaram, Andrea Caria, Ralf Veit, Tilman Gaber, Giuseppina Rota, Andrea Kuebler and Niels Birbaumer(2007) "FMRI Brain–Computer Interface: A Tool for Neuroscientific Research and Treatment"
- ^ Peplow, Mark (27 August 2004). "Mental ping-pong could aid paraplegics". News@nature. doi:10.1038/news040823-18.
- ^ "To operate robot only with brain, ATR and Honda develop BMI base technology". Tech-on. 26 May 2006. Archived from the original on 23 June 2017. Retrieved 22 September 2006.
- ^ Miyawaki Y, Uchida H, Yamashita O, Sato MA, Morito Y, Tanabe HC, et al. (December 2008). "Visual image reconstruction from human brain activity using a combination of multiscale local image decoders". Neuron. 60 (5): 915–929. doi:10.1016/j.neuron.2008.11.004. PMID 19081384. S2CID 17327816.
- ^ Nishimoto S, Vu AT, Naselaris T, Benjamini Y, Yu B, Gallant JL (October 2011). "Reconstructing visual experiences from brain activity evoked by natural movies". Current Biology. 21 (19): 1641–1646. doi:10.1016/j.cub.2011.08.031. PMC 3326357. PMID 21945275.
- ^ Yam, Philip (22 September 2011). "Breakthrough Could Enable Others to Watch Your Dreams and Memories". Scientific American. Retrieved 25 September 2011.
- ^ "Reconstructing visual experiences from brain activity evoked by natural movies (Project page)". The Gallant Lab at UC Berkeley. Archived from the original on 2011-09-25. Retrieved 25 September 2011.
- ^ Anwar, Yasmin (22 September 2011). "Scientists use brain imaging to reveal the movies in our mind". UC Berkeley News Center. Retrieved 25 September 2011.
- ^ a b c Marshall D, Coyle D, Wilson S, Callaghan M (2013). "Games, Gameplay, and BCI: The State of the Art". IEEE Transactions on Computational Intelligence and AI in Games. 5 (2): 83. doi:10.1109/TCIAIG.2013.2263555. S2CID 206636315.
- ^ "Goals of the organizers". BBC. Retrieved 19 December 2022.
- ^ Ang KK, Chin ZY, Wang C, Guan C, Zhang H (1 January 2012). "Filter Bank Common Spatial Pattern Algorithm on BCI Competition IV Datasets 2a and 2b". Frontiers in Neuroscience. 6: 39. doi:10.3389/fnins.2012.00039. PMC 3314883. PMID 22479236.
- ^ Fairclough, Stephen H. (January 2009). "Fundamentals of physiological computing". Interacting with Computers. 21 (1–2): 133–145. doi:10.1016/j.intcom.2008.10.011. S2CID 16314534.
- ^ Bozinovski S (2017). "Signal Processing Robotics Using Signals Generated by a Human Head: From Pioneering Works to EEG-Based Emulation of Digital Circuits". Advances in Robot Design and Intelligent Control. Advances in Intelligent Systems and Computing. Vol. 540. pp. 449–462. doi:10.1007/978-3-319-49058-8_49. ISBN 978-3-319-49057-1.
- ^ Mathôt S, Melmi JB, van der Linden L, Van der Stigchel S (2016). "The Mind-Writing Pupil: A Human-Computer Interface Based on Decoding of Covert Attention through Pupillometry". PLOS ONE. 11 (2): e0148805. Bibcode:2016PLoSO..1148805M. doi:10.1371/journal.pone.0148805. PMC 4743834. PMID 26848745.
- ^ Kennedy, Pagan (18 September 2011). "The Cyborg in Us All". The New York Times. Retrieved 28 January 2012.
- ^ Selim, Jocelyn; Drinkell, Pete (1 November 2002). "The Bionic Connection". Discover. Archived from the original on 6 January 2008.
- ^ Giaimo, Cara (10 June 2015). "Nervous System Hookup Leads to Telepathic Hand-Holding". Atlas Obscura.
- ^ Warwick, K, Gasson, M, Hutt, B, Goodhew, I, Kyberd, P, Schulzrinne, H and Wu, X: "Thought Communication and Control: A First Step using Radiotelegraphy", IEE Proceedings on Communications, 151(3), pp.185–189, 2004
- ^ Warwick K, Gasson M, Hutt B, Goodhew I, Kyberd P, Andrews B, et al. (October 2003). "The application of implant technology for cybernetic systems". Archives of Neurology. 60 (10): 1369–1373. doi:10.1001/archneur.60.10.1369. PMID 14568806.
- ^ Grau C, Ginhoux R, Riera A, Nguyen TL, Chauvat H, Berg M, et al. (2014). "Conscious brain-to-brain communication in humans using non-invasive technologies". PLOS ONE. 9 (8): e105225. Bibcode:2014PLoSO...9j5225G. doi:10.1371/journal.pone.0105225. PMC 4138179. PMID 25137064.
- ^ Bland, Eric (13 October 2008). "Army Developing 'synthetic telepathy'". Discovery News. Retrieved 13 October 2008.
- ^ Pais-Vieira M, Lebedev M, Kunicki C, Wang J, Nicolelis MA (28 February 2013). "A brain-to-brain interface for real-time sharing of sensorimotor information". Scientific Reports. 3: 1319. Bibcode:2013NatSR...3E1319P. doi:10.1038/srep01319. PMC 3584574. PMID 23448946.
- ^ Gorman, James (28 February 2013). "One Rat Thinks, and Another Reacts". The New York Times. Retrieved 28 February 2013.
- ^ Sample, Ian (1 March 2013). "Brain-to-brain interface lets rats share information via internet". The Guardian. Retrieved 2 March 2013.
- ^ Mazzatenta A, Giugliano M, Campidelli S, Gambazzi L, Businaro L, Markram H, et al. (June 2007). "Interfacing neurons with carbon nanotubes: electrical signal transfer and synaptic stimulation in cultured brain circuits". The Journal of Neuroscience. 27 (26): 6931–6936. doi:10.1523/JNEUROSCI.1051-07.2007. PMC 6672220. PMID 17596441.
- ^ Caltech Scientists Devise First Neurochip, Caltech, 26 October 1997
- ^ Sandhana, Lakshmi (22 October 2004). "Coming to a brain near you". Wired News. Archived from the original on 10 September 2006.
- ^ "'Brain' in a dish flies flight simulator". CNN. 4 November 2004.
- ^ "David Pearce – Humanity Plus". 2017-10-05. Retrieved 2021-12-30.
- ^ Stoica A (2010). "Speculations on Robots, Cyborgs & Telepresence". YouTube. Archived from the original on 28 December 2021. Retrieved 28 December 2021.
- ^ "Experts to 'redefine the future' at Humanity+ @ CalTech". Kurzweil. Retrieved 2021-12-30.
- ^ WO2012100081A2, Stoica, Adrian, "Aggregation of bio-signals from multiple individuals to achieve a collective outcome", issued 2012-07-26
- ^ Wang Y, Jung TP (2011-05-31). "A collaborative brain-computer interface for improving human performance". PLOS ONE. 6 (5): e20422. Bibcode:2011PLoSO...620422W. doi:10.1371/journal.pone.0020422. PMC 3105048. PMID 21655253.
- ^ Eckstein MP, Das K, Pham BT, Peterson MF, Abbey CK, Sy JL, Giesbrecht B (January 2012). "Neural decoding of collective wisdom with multi-brain computing". NeuroImage. 59 (1): 94–108. doi:10.1016/j.neuroimage.2011.07.009. PMID 21782959. S2CID 14930969.
- ^ Stoica A (September 2012). "MultiMind: Multi-Brain Signal Fusion to Exceed the Power of a Single Brain". 2012 Third International Conference on Emerging Security Technologies. pp. 94–98. doi:10.1109/EST.2012.47. ISBN 978-0-7695-4791-6. S2CID 6783719.
- ^ Poli R, Cinel C, Matran-Fernandez A, Sepulveda F, Stoica A (2013-03-19). "Towards cooperative brain-computer interfaces for space navigation". Proceedings of the 2013 international conference on Intelligent user interfaces. IUI '13. New York, NY, USA: Association for Computing Machinery. pp. 149–160. doi:10.1145/2449396.2449417. ISBN 978-1-4503-1965-2. S2CID 13201979.
- ^ Poli R, Cinel C, Sepulveda F, Stoica A (February 2013). "Improving decision-making based on visual perception via a collaborative brain-computer interface". 2013 IEEE International Multi-Disciplinary Conference on Cognitive Methods in Situation Awareness and Decision Support (CogSIMA). San Diego, CA: IEEE. pp. 1–8. doi:10.1109/CogSIMA.2013.6523816. ISBN 978-1-4673-2437-3. S2CID 25136642.
- ^ Matran-Fernandez A, Poli R, Cinel C (November 2013). "Collaborative brain-computer interfaces for the automatic classification of images". 2013 6th International IEEE/EMBS Conference on Neural Engineering (NER). pp. 1096–1099. doi:10.1109/NER.2013.6696128. ISBN 978-1-4673-1969-0. S2CID 40341170.
- ^ Valeriani D, Cinel C, Poli R (August 2017). "Group Augmentation in Realistic Visual-Search Decisions via a Hybrid Brain-Computer Interface". Scientific Reports. 7 (1): 7772. Bibcode:2017NatSR...7.7772V. doi:10.1038/s41598-017-08265-7. PMC 5552884. PMID 28798411.
- ^ Bhattacharyya S, Valeriani D, Cinel C, Citi L, Poli R (August 2021). "Anytime collaborative brain-computer interfaces for enhancing perceptual group decision-making". Scientific Reports. 11 (1): 17008. Bibcode:2021NatSR..1117008B. doi:10.1038/s41598-021-96434-0. PMC 8379268. PMID 34417494.
- ^ "Paralyzed Again". MIT Technology Review. Retrieved 2023-12-08.
- ^ "Gale - Product Login". galeapps.gale.com. Retrieved 2023-12-08.
- ^ Ienca, Marcello; Haselager, Pim (June 2016). "Hacking the brain: brain-computer interfacing technology and the ethics of neurosecurity". Ethics & Information Technology. 18 (2): 117–129. doi:10.1007/s10676-016-9398-9. S2CID 5132634.
- ^ Steinert, Steffen; Friedrich, Orsolya (2020-02-01). "Wired Emotions: Ethical Issues of Affective Brain–Computer Interfaces". Science and Engineering Ethics. 26 (1): 351–367. doi:10.1007/s11948-019-00087-2. ISSN 1471-5546. PMC 6978299. PMID 30868377.
- ^ Clausen, Jens (2009-02-01). "Man, machine and in between". Nature. 457 (7233): 1080–1081. Bibcode:2009Natur.457.1080C. doi:10.1038/4571080a. ISSN 0028-0836. PMID 19242454. S2CID 205043226.
- ^ Haselager, Pim; Vlek, Rutger; Hill, Jeremy; Nijboer, Femke (2009-11-01). "A note on ethical aspects of BCI". Neural Networks. Brain-Machine Interface. 22 (9): 1352–1357. doi:10.1016/j.neunet.2009.06.046. hdl:2066/77533. ISSN 0893-6080. PMID 19616405.
- ^ Attiah, Mark A.; Farah, Martha J. (2014-05-15). "Minds, motherboards, and money: futurism and realism in the neuroethics of BCI technologies". Frontiers in Systems Neuroscience. 8: 86. doi:10.3389/fnsys.2014.00086. ISSN 1662-5137. PMC 4030132. PMID 24860445.
- ^ Nijboer, Femke; Clausen, Jens; Allison, Brendan Z.; Haselager, Pim (2013). "The Asilomar Survey: Stakeholders' Opinions on Ethical Issues Related to Brain-Computer Interfacing". Neuroethics. 6 (3): 541–578. doi:10.1007/s12152-011-9132-6. ISSN 1874-5490. PMC 3825606. PMID 24273623.
- ^ "Sony patent neural interface". Archived from the original on 7 April 2012.
- ^ "Mind Games". The Economist. 23 March 2007.
- ^ "nia Game Controller Product Page". OCZ Technology Group. Retrieved 30 January 2013.
- ^ a b c Li S (8 August 2010). "Mind reading is on the market". Los Angeles Times. Archived from the original on 4 January 2013.
- ^ Fruhlinger, Joshua (9 October 2008). "Brains-on with NeuroSky and Square Enix's Judecca mind-control game". Engadget. Retrieved 29 May 2012.
- ^ New games powered by brain waves. Physorg.com (10 January 2009). Retrieved on 12 September 2010.
- ^ Snider, Mike (7 January 2009). "Toy trains 'Star Wars' fans to use The Force". USA Today. Retrieved 1 May 2010.
- ^ "Emotiv Homepage". Emotiv.com. Retrieved 29 December 2009.
- ^ "'necomimi' selected 'Time Magazine / The 50 best invention of the year'". Neurowear. 22 November 2011. Archived from the original on 25 January 2012.
- ^ "LIFESUIT Updates & News – They Shall Walk". Theyshallwalk.org. Retrieved 19 December 2016.
- ^ "SmartphoneBCI". GitHub. Retrieved 5 June 2018.
- ^ "SSVEP_keyboard". GitHub. Retrieved 5 April 2017.
- ^ Protalinski, Emil (8 December 2020). "NextMind ships its real-time brain computer interface Dev Kit for $399". VentureBeat. Retrieved 8 September 2021.
- ^ Etherington, Darrell (21 December 2020). "NextMind's Dev Kit for mind-controlled computing offers a rare 'wow' factor in tech". TechCrunch. Retrieved 1 April 2024.
- ^ "Roadmap - BNCI Horizon 2020". bnci-horizon-2020.eu. Retrieved 2019-05-05.
- ^ Brunner C, Birbaumer N, Blankertz B, Guger C, Kübler A, Mattia D, et al. (2015). "BNCI Horizon 2020: towards a roadmap for the BCI community". Brain-Computer Interfaces. 2: 1–10. doi:10.1080/2326263X.2015.1008956. hdl:1874/350349. S2CID 15822773.
- ^ Allison BZ, Dunne S, Leeb R, Millan J, Nijholt A (2013). Towards Practical Brain-Computer Interfaces: Bridging the Gap from Research to Real-World Applications. Berlin Heidelberg: Springer Verlag. ISBN 978-3-642-29746-5.
- ^ Edlinger G, Allison BZ, Guger C (2015). "How many people could use a BCI system?". In Kansaku K, Cohen L, Birbaumer N (eds.). Clinical Systems Neuroscience. Tokyo: pringer Verlag Japan. pp. 33–66. ISBN 978-4-431-55037-2.
- ^ Chatelle C, Chennu S, Noirhomme Q, Cruse D, Owen AM, Laureys S (2012). "Brain-computer interfacing in disorders of consciousness". Brain Injury. 26 (12): 1510–1522. doi:10.3109/02699052.2012.698362. hdl:2268/162403. PMID 22759199. S2CID 6498232.
- ^ Boly M, Massimini M, Garrido MI, Gosseries O, Noirhomme Q, Laureys S, Soddu A (2012). "Brain connectivity in disorders of consciousness". Brain Connectivity. 2 (1): 1–10. doi:10.1089/brain.2011.0049. hdl:2268/131984. PMID 22512333. S2CID 6447538.
- ^ Gibson RM, Fernández-Espejo D, Gonzalez-Lara LE, Kwan BY, Lee DH, Owen AM, Cruse D (2014). "Multiple tasks and neuroimaging modalities increase the likelihood of detecting covert awareness in patients with disorders of consciousness". Frontiers in Human Neuroscience. 8: 950. doi:10.3389/fnhum.2014.00950. PMC 4244609. PMID 25505400.
- ^ Risetti M, Formisano R, Toppi J, Quitadamo LR, Bianchi L, Astolfi L, et al. (2013). "On ERPs detection in disorders of consciousness rehabilitation". Frontiers in Human Neuroscience. 7: 775. doi:10.3389/fnhum.2013.00775. PMC 3834290. PMID 24312041.
- ^ Silvoni S, Ramos-Murguialday A, Cavinato M, Volpato C, Cisotto G, Turolla A, et al. (October 2011). "Brain-computer interface in stroke: a review of progress". Clinical EEG and Neuroscience. 42 (4): 245–252. doi:10.1177/155005941104200410. PMID 22208122. S2CID 37902399.
- ^ Leamy DJ, Kocijan J, Domijan K, Duffin J, Roche RA, Commins S, et al. (January 2014). "An exploration of EEG features during recovery following stroke - implications for BCI-mediated neurorehabilitation therapy". Journal of Neuroengineering and Rehabilitation. 11: 9. doi:10.1186/1743-0003-11-9. PMC 3996183. PMID 24468185.
- ^ Tung SW, Guan C, Ang KK, Phua KS, Wang C, Zhao L, et al. (July 2013). "Motor imagery BCI for upper limb stroke rehabilitation: An evaluation of the EEG recordings using coherence analysis". 2013 35th Annual International Conference of the IEEE Engineering in Medicine and Biology Society (EMBC). Vol. 2013. pp. 261–264. doi:10.1109/EMBC.2013.6609487. ISBN 978-1-4577-0216-7. PMID 24109674. S2CID 5071115.
- ^ Bai Z, Fong KN, Zhang JJ, Chan J, Ting KH (April 2020). "Immediate and long-term effects of BCI-based rehabilitation of the upper extremity after stroke: a systematic review and meta-analysis". Journal of Neuroengineering and Rehabilitation. 17 (1): 57. doi:10.1186/s12984-020-00686-2. PMC 7183617. PMID 32334608.
- ^ Remsik A, Young B, Vermilyea R, Kiekhoefer L, Abrams J, Evander Elmore S, et al. (May 2016). "A review of the progression and future implications of brain-computer interface therapies for restoration of distal upper extremity motor function after stroke". Expert Review of Medical Devices. 13 (5): 445–454. doi:10.1080/17434440.2016.1174572. PMC 5131699. PMID 27112213.
- ^ Monge-Pereira E, Ibañez-Pereda J, Alguacil-Diego IM, Serrano JI, Spottorno-Rubio MP, Molina-Rueda F (September 2017). "Use of Electroencephalography Brain-Computer Interface Systems as a Rehabilitative Approach for Upper Limb Function After a Stroke: A Systematic Review". PM&R. 9 (9): 918–932. doi:10.1016/j.pmrj.2017.04.016. PMID 28512066. S2CID 20808455.
- ^ Sabathiel N, Irimia DC, Allison BZ, Guger C, Edlinger G (17 July 2016). "Paired Associative Stimulation with Brain-Computer Interfaces: A New Paradigm for Stroke Rehabilitation". Foundations of Augmented Cognition: Neuroergonomics and Operational Neuroscience. Lecture Notes in Computer Science. Vol. 9743. pp. 261–272. doi:10.1007/978-3-319-39955-3_25. ISBN 978-3-319-39954-6.
- ^ Riccio A, Pichiorri F, Schettini F, Toppi J, Risetti M, Formisano R, et al. (2016). "Interfacing brain with computer to improve communication and rehabilitation after brain damage". Brain-Computer Interfaces: Lab Experiments to Real-World Applications. Progress in Brain Research. Vol. 228. pp. 357–387. doi:10.1016/bs.pbr.2016.04.018. ISBN 978-0-12-804216-8. PMID 27590975.
- ^ Várkuti B, Guan C, Pan Y, Phua KS, Ang KK, Kuah CW, et al. (January 2013). "Resting state changes in functional connectivity correlate with movement recovery for BCI and robot-assisted upper-extremity training after stroke". Neurorehabilitation and Neural Repair. 27 (1): 53–62. doi:10.1177/1545968312445910. PMID 22645108. S2CID 7120989.
- ^ Young BM, Nigogosyan Z, Remsik A, Walton LM, Song J, Nair VA, et al. (2014). "Changes in functional connectivity correlate with behavioral gains in stroke patients after therapy using a brain-computer interface device". Frontiers in Neuroengineering. 7: 25. doi:10.3389/fneng.2014.00025. PMC 4086321. PMID 25071547.
- ^ a b Yuan K, Chen C, Wang X, Chu WC, Tong RK (January 2021). "BCI Training Effects on Chronic Stroke Correlate with Functional Reorganization in Motor-Related Regions: A Concurrent EEG and fMRI Study". Brain Sciences. 11 (1): 56. doi:10.3390/brainsci11010056. PMC 7824842. PMID 33418846.
- ^ Mrachacz-Kersting N, Voigt M, Stevenson AJ, Aliakbaryhosseinabadi S, Jiang N, Dremstrup K, Farina D (November 2017). "The effect of type of afferent feedback timed with motor imagery on the induction of cortical plasticity". Brain Research. 1674: 91–100. doi:10.1016/j.brainres.2017.08.025. hdl:10012/12325. PMID 28859916. S2CID 5866337.
- ^ Opie N (2 April 2019). "Research Overview". University of Melbourne Medicine. University of Melbourne. Retrieved 5 December 2019.
- ^ Oxley TJ, Opie NL, John SE, Rind GS, Ronayne SM, Wheeler TL, et al. (March 2016). "Minimally invasive endovascular stent-electrode array for high-fidelity, chronic recordings of cortical neural activity". Nature Biotechnology. 34 (3): 320–327. doi:10.1038/nbt.3428. PMID 26854476. S2CID 205282364.
- ^ "Synchron begins trialling Stentrode neural interface technology". Verdict Medical Devices. 22 September 2019. Retrieved 5 December 2019.
- ^ Radzik I, Miziak B, Dudka J, Chrościńska-Krawczyk M, Czuczwar SJ (June 2015). "Prospects of epileptogenesis prevention". Pharmacological Reports. 67 (3): 663–668. doi:10.1016/j.pharep.2015.01.016. PMID 25933984. S2CID 31284248.
- ^ Ritaccio A, Brunner P, Gunduz A, Hermes D, Hirsch LJ, Jacobs J, et al. (December 2014). "Proceedings of the Fifth International Workshop on Advances in Electrocorticography". Epilepsy & Behavior. 41: 183–192. doi:10.1016/j.yebeh.2014.09.015. PMC 4268064. PMID 25461213.
- ^ Kim DH, Viventi J, Amsden JJ, Xiao J, Vigeland L, Kim YS, et al. (June 2010). "Dissolvable films of silk fibroin for ultrathin conformal bio-integrated electronics". Nature Materials. 9 (6): 511–517. Bibcode:2010NatMa...9..511K. doi:10.1038/nmat2745. PMC 3034223. PMID 20400953.
- ^ Boppart SA, Wheeler BC, Wallace CS (January 1992). "A flexible perforated microelectrode array for extended neural recordings". IEEE Transactions on Bio-Medical Engineering. 39 (1): 37–42. doi:10.1109/10.108125. PMID 1572679. S2CID 36593459.
- ^ Kim DH, Ghaffari R, Lu N, Rogers JA (2012). "Flexible and stretchable electronics for biointegrated devices". Annual Review of Biomedical Engineering. 14: 113–128. doi:10.1146/annurev-bioeng-071811-150018. PMID 22524391. S2CID 5223203.
- ^ Thompson CH, Zoratti MJ, Langhals NB, Purcell EK (April 2016). "Regenerative Electrode Interfaces for Neural Prostheses". Tissue Engineering. Part B, Reviews. 22 (2): 125–135. doi:10.1089/ten.teb.2015.0279. PMID 26421660.
- ^ a b Rabaey JM (September 2011). "Brain-machine interfaces as the new frontier in extreme miniaturization". 2011 Proceedings of the European Solid-State Device Research Conference (ESSDERC). pp. 19–24. doi:10.1109/essderc.2011.6044240. ISBN 978-1-4577-0707-0. S2CID 47542923.
- ^ Warneke B, Last M, Liebowitz B, Pister KS (January 2001). "Smart Dust: communicating with a cubic-millimeter computer". Computer. 34 (1): 44–51. doi:10.1109/2.895117. ISSN 0018-9162. S2CID 21557.
Further reading[edit]
- Brouse, Andrew. "A Young Person's Guide to Brainwave Music: Forty years of audio from the human EEG". eContact! 14.2 – Biotechnological Performance Practice / Pratiques de performance biotechnologique (July 2012). Montréal: CEC.
- Gupta, Cota Navin and Ramaswamy Palanappian. "Using High-Frequency Electroencephalogram in Visual and Auditory-Based Brain-Computer Interface Designs". eContact! 14.2 – Biotechnological Performance Practice / Pratiques de performance biotechnologique (July 2012). Montréal: CEC.
- Ouzounian, Gascia. "The Biomuse Trio in Conversation: An Interview with R. Benjamin Knapp and Eric Lyon". eContact! 14.2 – Biotechnological Performance Practice / Pratiques de performance biotechnologique (July 2012). Montréal: CEC.
External links[edit]