User:Mr. Ibrahem/Amoxicillin/clavulanic acid
 | |
 | |
| Combination of | |
|---|---|
| Amoxicillin | Penicillin antibiotic |
| Clavulanic acid | Beta-lactamase inhibitor |
| Clinical data | |
| Trade names | Augmentin, Clavulin, other[1] |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a685024 |
| License data | |
| Pregnancy category |
|
| Routes of administration | By mouth, intravenous[2] |
| Drug class | Antibiotic |
| Legal status | |
| Legal status | |
| Chemical and physical data | |
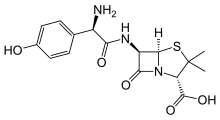
| Formula | C24H27KN4O10S |
| Molar mass | 602.66 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
| | |
Amoxicillin/clavulanic acid, also known as co-amoxiclav, is an antibiotic useful for the treatment of a number of bacterial infections.[3] It is a combination consisting of amoxicillin, a β-lactam antibiotic, and potassium clavulanate, a β-lactamase inhibitor.[3] It is specifically used for otitis media, strep throat, pneumonia, cellulitis, urinary tract infections, and animal bites.[3] It is taken by mouth or by injection into a vein.[2]
Common side effects include diarrhea, vomiting, and allergic reactions.[3] It also increases the risk of yeast infections, headaches, and blood clotting problems.[2][5] It is not recommended in people with a history of a penicillin allergy.[2] It is relatively safe for use during pregnancy.[3]
Amoxicillin/clavulanic acid was approved for medical use in the United States in 1984.[3] It is on the World Health Organization's List of Essential Medicines.[6] It is available as a generic medication.[3] The wholesale cost in the developing world is about US$0.18–1.14 per day.[7] In the United States a course of treatment costs $50–100.[1] In 2017, it was the 116th most commonly prescribed medication in the United States, with more than six million prescriptions.[8][9]
References[edit]
- ^ a b Hamilton, Richart (2015). Tarascon Pocket Pharmacopoeia 2015 Deluxe Lab-Coat Edition. Jones & Bartlett Learning. p. 97. ISBN 9781284057560.
- ^ a b c d World Health Organization (2009). Stuart MC, Kouimtzi M, Hill SR (eds.). WHO Model Formulary 2008. World Health Organization. p. 102. hdl:10665/44053. ISBN 9789241547659.
- ^ a b c d e f g h i "Amoxicillin and Clavulanate Potassium". The American Society of Health-System Pharmacists. Archived from the original on 29 November 2016. Retrieved 8 December 2016.
- ^ a b "WHOCC - ATC/DDD Index". www.whocc.no. Archived from the original on 31 May 2020. Retrieved 1 September 2020.
- ^ Gillies, M; Ranakusuma, A; Hoffmann, T; Thorning, S; McGuire, T; Glasziou, P; Del Mar, C (17 November 2014). "Common harms from amoxicillin: a systematic review and meta-analysis of randomized placebo-controlled trials for any indication". Canadian Medical Association Journal. 187: E21-31. doi:10.1503/cmaj.140848. PMC 4284189. PMID 25404399.
- ^ World Health Organization (2019). World Health Organization model list of essential medicines: 21st list 2019. Geneva: World Health Organization. hdl:10665/325771. WHO/MVP/EMP/IAU/2019.06. License: CC BY-NC-SA 3.0 IGO.
- ^ "Amoxicillin + Clavulanic Acid". International Drug Price Indicator Guide. Archived from the original on 22 January 2018. Retrieved 8 December 2016.
- ^ "The Top 300 of 2020". ClinCalc. Archived from the original on 18 March 2020. Retrieved 11 April 2020.
- ^ "Amoxicillin; Clavulanate Potassium - Drug Usage Statistics". ClinCalc. Archived from the original on 11 April 2020. Retrieved 11 April 2020.
